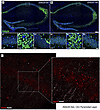
Decoding muscle-resident Schwann cell dynamics during neuromuscular junction remodeling
Guzman et al. report single-cell RNA sequencing data that reveal how distinct Schwann cell subtypes and SPP1 signaling coordinate neuromuscular junction (NMJ) repair following partial and complete muscle denervation in mice. The cover shows muscle fiber imaging from a mouse model of NMJ degeneration and denervation that expresses a Schwann cell reporter, with staining for S100B (green), Ki67 (magenta), α-bungarotoxin (red), and DAPI (blue).
-
Review
Yohannes Tesfaigzi, Ali Önder Yildirim, Francesca Polverino, Thomas M. Conlon, Venkataramana Sidhaye, Maor Sauler, S. Vamsee Raju, Renata Z. Jurkowska, Divay Chandra, Michael H. Cho, Edwin K. Silverman, Ramon C. Sun, Peter Castaldi, Purushothama Rao Tata, Kambez H. Benam, Linto Antony, Mareike Lehmann, Beata Kosmider, Karim Bahmed, Zerihun H. Negasi, Kamakshi Bankoti, Carter Swaby, Dave A. Lagowala, Yeşim Vural, Hasan Bayram, Rosa Faner, George Washko, Dinh Son Bui, Bartolome Celli, Roxana Maria Wasnick, Enid NeptuneYohannes Tesfaigzi, Ali Önder Yildirim, Francesca Polverino, Thomas M. Conlon, Venkataramana Sidhaye, Maor Sauler, S. Vamsee Raju, Renata Z. Jurkowska, Divay Chandra, Michael H. Cho, Edwin K. Silverman, Ramon C. Sun, Peter Castaldi, Purushothama Rao Tata, Kambez H. Benam, Linto Antony, Mareike Lehmann, Beata Kosmider, Karim Bahmed, Zerihun H. Negasi, Kamakshi Bankoti, Carter Swaby, Dave A. Lagowala, Yeşim Vural, Hasan Bayram, Rosa Faner, George Washko, Dinh Son Bui, Bartolome Celli, Roxana Maria Wasnick, Enid Neptune×
Abstract
Chronic obstructive pulmonary disease (COPD) was the third leading cause of global mortality in 2011 but receives limited attention and research funding. This Review describes the current knowledge on COPD risk factors, including genetic and epigenetic determinants and their interactions with the microbiome and environmental exposures. Preclinical models are being refined and single-cell transcriptomic, metabolomic, and proteomic technologies are being implemented to investigate the molecular mechanisms of disease progression. Patient cohorts to define biomarkers of early disease and the latest approaches to diagnose pre-COPD are essential to accelerate the development of novel and effective therapeutic interventions and translate new findings into clinical trials. This Review is a summary of topics covered by a symposium organized by the COPD-iNET consortium, an international network of researchers who have established a platform that facilitates collaboration of this multidisciplinary group of preclinical, translational, and clinical researchers.
Authors
Yohannes Tesfaigzi, Ali Önder Yildirim, Francesca Polverino, Thomas M. Conlon, Venkataramana Sidhaye, Maor Sauler, S. Vamsee Raju, Renata Z. Jurkowska, Divay Chandra, Michael H. Cho, Edwin K. Silverman, Ramon C. Sun, Peter Castaldi, Purushothama Rao Tata, Kambez H. Benam, Linto Antony, Mareike Lehmann, Beata Kosmider, Karim Bahmed, Zerihun H. Negasi, Kamakshi Bankoti, Carter Swaby, Dave A. Lagowala, Yeşim Vural, Hasan Bayram, Rosa Faner, George Washko, Dinh Son Bui, Bartolome Celli, Roxana Maria Wasnick, Enid Neptune
-
Research Articles
Pujun Guan, Zihang Chen, Hanze Dong, Xia Guo, Juan Huang, Tian Dong, Mi Wang, Xiaoxi Lu, Fei Huang, Wenbin Li, Yuan Tang, Li Zhang, Ling Pan, Ju Gao, Shikun Wang, Rongbo Liu, Wenyan Zhang, Sha Zhao, Weiping LiuPujun Guan, Zihang Chen, Hanze Dong, Xia Guo, Juan Huang, Tian Dong, Mi Wang, Xiaoxi Lu, Fei Huang, Wenbin Li, Yuan Tang, Li Zhang, Ling Pan, Ju Gao, Shikun Wang, Rongbo Liu, Wenyan Zhang, Sha Zhao, Weiping Liu×
Abstract
Systemic Epstein-Barr virus–positive (EBV-positive) T/NK cell lymphoproliferative diseases of childhood (sEBV+T/NK-LPD) are a spectrum of rare diseases that have highly variable biological behavior, from indolent conditions to highly aggressive malignancies. Clinicians currently face substantial challenges in promptly assessing disease severity and predicting patient outcomes, leading to limitations in treatment planning. To address this challenge, we constructed a comprehensive triage system to aid in rapid clinical interventions. The study included 156 patients with newly diagnosed sEBV+T/NK-LPD from 42 institutions. An independent prospective cohort of 35 newly enrolled patients was further included to evaluate the model’s performance. An additional 45 patients from the literature and 18 patients who underwent hematopoietic stem cell transplantation were included to test the score’s generalizability. An integrative machine learning strategy was applied to identify robust and optimal factors and to integrate multiple algorithms to enhance the system’s performance and stability. This system, termed COLLAPSED, identifies critical factors and provides a stable, high-performing ensemble. This model was validated externally and simplified into a risk score to improve interpretability and accessibility. The COLLAPSED system substantially enhances clinicians’ ability to rapidly and precisely identify high-risk patients, thus enabling timely clinical decision-making and expedited initiation of potentially lifesaving treatments.
Authors
Pujun Guan, Zihang Chen, Hanze Dong, Xia Guo, Juan Huang, Tian Dong, Mi Wang, Xiaoxi Lu, Fei Huang, Wenbin Li, Yuan Tang, Li Zhang, Ling Pan, Ju Gao, Shikun Wang, Rongbo Liu, Wenyan Zhang, Sha Zhao, Weiping Liu
×Abstract
Huntington’s disease (HD) is a neurodegenerative disorder caused by the expansion of CAG repeats in the gene encoding huntingtin. Since accumulation of mutant huntingtin (mHtt) leads to dysfunction of numerous cellular pathways and toxicity, reducing levels of the mutant protein represents a key therapeutic objective in HD. We found that ubiquitination of mHtt by E3 ubiquitin ligase Nedd4 promotes clearance of the mutant protein. Knockdown of Nedd4 increased toxicity of mHtt in mouse primary neurons and in a fly model of HD, suggesting the protective role of Nedd4. Importantly, levels of Nedd4 were decreased in mHtt-expressing neurons through impaired mTORC1 activity, suggesting a feedback loop of mHtt accumulation and Nedd4 reduction that leads to accumulation and, ultimately, toxicity of mHtt. These findings suggest that restoring Nedd4 activity may offer a novel therapeutic opportunity for HD.
Authors
Hyunkyung Jeong, Yiyang Qin, Fangke Xu, Katarina Trajkovic, Myung Jong Kim, Nicolas Marotta, Kana Hamada, Ravi Allada, Su Yang, Dimitri Krainc
Wen Gu, Nicole Wilkinson, Carine Fillebeen, Darren M. Blackburn, Korin Sahinyan, Eric Bonneil, Tao Zhao, Zhi Luo, Vahab D. Soleimani, Vincent Richard, Christoph H. Borchers, Albert Koulman, Benjamin Jenkins, Bernhard Michalke, Hans Zischka, Judith Sailer, Vivek Venkataramani, Othon Iliopoulos, Gary Sweeney, Kostas PantopoulosWen Gu, Nicole Wilkinson, Carine Fillebeen, Darren M. Blackburn, Korin Sahinyan, Eric Bonneil, Tao Zhao, Zhi Luo, Vahab D. Soleimani, Vincent Richard, Christoph H. Borchers, Albert Koulman, Benjamin Jenkins, Bernhard Michalke, Hans Zischka, Judith Sailer, Vivek Venkataramani, Othon Iliopoulos, Gary Sweeney, Kostas Pantopoulos×Abstract
Iron regulatory protein 1 (IRP1) is a posttranscriptional regulator of cellular iron metabolism. In mice, loss of IRP1 causes polycythemia through translational de-repression of HIF2α mRNA, which increases renal erythropoietin production. Here, we show that Irp1–/– mice develop fasting hypoglycemia and are protected against high-fat diet–induced hyperglycemia and hepatic steatosis. Discovery-based proteomics of Irp1–/– livers revealed a mitochondrial dysfunction signature. Seahorse flux analysis in primary hepatocytes and differentiated skeletal muscle myotubes confirmed impaired respiratory capacity, with a shift from oxidative phosphorylation to glycolytic ATP production. This metabolic rewiring was associated with enhanced insulin sensitivity and increased glucose uptake in skeletal muscle. Under metabolic stress, IRP1 deficiency altered the redox balance of mitochondrial iron, resulting in inefficient energy production and accumulation of amino acids and metabolites in skeletal muscles, rendering them unavailable for hepatic gluconeogenesis. These findings identify IRP1 as a critical regulator of systemic energy homeostasis.
Authors
Wen Gu, Nicole Wilkinson, Carine Fillebeen, Darren M. Blackburn, Korin Sahinyan, Eric Bonneil, Tao Zhao, Zhi Luo, Vahab D. Soleimani, Vincent Richard, Christoph H. Borchers, Albert Koulman, Benjamin Jenkins, Bernhard Michalke, Hans Zischka, Judith Sailer, Vivek Venkataramani, Othon Iliopoulos, Gary Sweeney, Kostas Pantopoulos
Nora Yucel, Silvia Marchiano, Evan Tchelepi, Germana Paterlini, Ivan A. Kuznetsov, Kristina Li, Quentin McAfee, Nehaar Nimmagadda, Andy Ren, Sam Shi, Alyssa Grogan, Aikaterini Kontrogianni-Konstantopoulos, Charles Murry, Zoltan AranyNora Yucel, Silvia Marchiano, Evan Tchelepi, Germana Paterlini, Ivan A. Kuznetsov, Kristina Li, Quentin McAfee, Nehaar Nimmagadda, Andy Ren, Sam Shi, Alyssa Grogan, Aikaterini Kontrogianni-Konstantopoulos, Charles Murry, Zoltan Arany×Abstract
How SARS-CoV-2 causes a wide range of clinical manifestations and disease severity remains poorly understood. SARS-CoV-2 encodes 2 proteases (3CLPro and PLPro), vital for viral production, but also promiscuous with respect to host protein targets. Pharmacological inhibition of 3CLPro markedly reduced hospitalization and death in Phase 2/3 clinical studies. Here, we develop a bioinformatic algorithm, leveraging experimental data from SARS-CoV, to predict host cleavage targets of 3CLPro. We capture targets of 3CLPro described previously for SARS-CoV-2, as well as thousands of putative targets. We validate numerous targets cleaved during infection, including the giant sarcomeric protein obscurin and the innate immune protein OAS1. A long form of OAS1, p46, has been associated in numerous GWAS studies with lesser COVID disease severity. We show that 3CLPro cleaves p46 OAS1 immediately upstream of a known prenylation domain, relocalizing OAS1 from subcellular membranes to the cytosol, rendering it akin to the nonprotective, cytosolic p42 isoform. Similar OAS1 relocalization occurs upon infection by SARS-CoV-2. Our data provide a high-throughput resource to identify putative host cleavage targets of 3CLPro and reveal a mechanism by which SARS-CoV-2 antagonizes host innate immunity in individuals with the protective p46 isoform of OAS1.
Authors
Nora Yucel, Silvia Marchiano, Evan Tchelepi, Germana Paterlini, Ivan A. Kuznetsov, Kristina Li, Quentin McAfee, Nehaar Nimmagadda, Andy Ren, Sam Shi, Alyssa Grogan, Aikaterini Kontrogianni-Konstantopoulos, Charles Murry, Zoltan Arany
Nikolaos T. Skenteris, Elisa Luttermann, Sanjana Nair, Ioannis Evangelakos, Maria Pujantell, Marie Eggers, Fabian Hausmann, Marleen Bérouti, Benedetta Padoan, Felix J. Flomm, Janna M. Claussen, Benjamin Grünhagel, Anika Salfelder, Brigitte Beifuss, Saskia Biskup, Patrick Blümke, Katrin Rading, Heike Hildebrandt, Urte Matschl, Silke Giesemann-Jansen, Jana Hennesen, Viacheslav O. Nikolaev, Michael Kutsche, Christian Kubisch, Friedrich Koch-Nolte, Nicola M. Tomas, Eva Tolosa, Marc Lütgehetmann, Felix R. Stahl, Veit Hornung, Madeleine J. Bunders, Christian Schlein, Maya Topf, Ina Kötter, Marcus AltfeldNikolaos T. Skenteris, Elisa Luttermann, Sanjana Nair, Ioannis Evangelakos, Maria Pujantell, Marie Eggers, Fabian Hausmann, Marleen Bérouti, Benedetta Padoan, Felix J. Flomm, Janna M. Claussen, Benjamin Grünhagel, Anika Salfelder, Brigitte Beifuss, Saskia Biskup, Patrick Blümke, Katrin Rading, Heike Hildebrandt, Urte Matschl, Silke Giesemann-Jansen, Jana Hennesen, Viacheslav O. Nikolaev, Michael Kutsche, Christian Kubisch, Friedrich Koch-Nolte, Nicola M. Tomas, Eva Tolosa, Marc Lütgehetmann, Felix R. Stahl, Veit Hornung, Madeleine J. Bunders, Christian Schlein, Maya Topf, Ina Kötter, Marcus Altfeld×Abstract
With the increasing use of genetic sequencing to investigate inborn errors of immunity, rare variants are frequently identified, yet their clinical relevance often remains uncertain. Establishing pathogenicity requires a multidisciplinary approach that integrates genetic, structural, functional, and clinical data. Here, we used such a strategy to investigate a previously unreported hemizygous missense variant — alanine (A) to threonine (T) at residue 518 — in Toll-like receptor 8 (TLR8), identified in 2 male siblings with recurrent infections and systemic inflammation, characterized by a proinflammatory immune signature and B cell dysregulation. Functional studies showed that the TLR8 A518T variant enhanced NF-κB activation and increased secretion of proinflammatory cytokines compared with WT TLR8 upon stimulation, consistent with a gain-of-function effect. Protein degradation and turnover assays revealed reduced abundance of the mutant TLR8 protein due to faster turnover and increased proteasomal degradation. Computational modeling predicted enhanced structural stabilization of the active TLR8 homodimer interface via additional water-mediated hydrogen bonds introduced by the A518T substitution. Together, these findings integrating structural modeling with functional assays identify a novel TLR8 ligand-specific gain-of-function mutation resulting in complex immunopathology in 2 siblings.
Authors
Nikolaos T. Skenteris, Elisa Luttermann, Sanjana Nair, Ioannis Evangelakos, Maria Pujantell, Marie Eggers, Fabian Hausmann, Marleen Bérouti, Benedetta Padoan, Felix J. Flomm, Janna M. Claussen, Benjamin Grünhagel, Anika Salfelder, Brigitte Beifuss, Saskia Biskup, Patrick Blümke, Katrin Rading, Heike Hildebrandt, Urte Matschl, Silke Giesemann-Jansen, Jana Hennesen, Viacheslav O. Nikolaev, Michael Kutsche, Christian Kubisch, Friedrich Koch-Nolte, Nicola M. Tomas, Eva Tolosa, Marc Lütgehetmann, Felix R. Stahl, Veit Hornung, Madeleine J. Bunders, Christian Schlein, Maya Topf, Ina Kötter, Marcus Altfeld
Ahmed A. Rahman, Rhian Stavely, Leah C. Ott, Christopher Y. Han, Kensuke Ohishi, Ryo Hotta, Alan J. Burns, Sabyasachi Das, Emily Da Cruz, Diana Tambala, Mark E. Lindsay, Patricia L. Musolino, Allan M. GoldsteinAhmed A. Rahman, Rhian Stavely, Leah C. Ott, Christopher Y. Han, Kensuke Ohishi, Ryo Hotta, Alan J. Burns, Sabyasachi Das, Emily Da Cruz, Diana Tambala, Mark E. Lindsay, Patricia L. Musolino, Allan M. Goldstein×Abstract
Multisystemic smooth muscle dysfunction syndrome (MSMDS) is a rare disorder caused by ACTA2 mutations, including the R179H variant, which alters actin filament stability and dynamics and smooth muscle contractility. Cardiovascular complications dominate its clinical presentation, but gastrointestinal (GI) dysfunction significantly affects quality of life. To investigate the structural, functional, and cellular basis of gut dysmotility in MSMDS, we reviewed clinical data from 24 patients with MSMDS and studied the ACTA2 R179H mouse model. Patients exhibited severe gut dysmotility, with 75% requiring medication for chronic constipation. ACTA2 mutant mice displayed cecal and colonic dilatation, reduced intestinal length, and disrupted colonic migrating motor complexes. Delayed whole-gut transit and impaired contractile responses to electrical and pharmacological stimulation were observed. Transcriptomic analysis revealed significant actin cytoskeleton-related gene changes in smooth muscle cells, and immune profiling identified increased lymphocytic infiltration. Despite functional abnormalities, there were no obvious changes in the enteric nervous system. These findings establish ACTA2 mice as a robust model for studying GI pathology in MSMDS, elucidating the role of smooth muscle dysfunction in gut dysmotility. This model provides a foundation for developing targeted therapies aimed at restoring intestinal motility by directly addressing actin cytoskeletal disruptions in smooth muscle cells.
Authors
Ahmed A. Rahman, Rhian Stavely, Leah C. Ott, Christopher Y. Han, Kensuke Ohishi, Ryo Hotta, Alan J. Burns, Sabyasachi Das, Emily Da Cruz, Diana Tambala, Mark E. Lindsay, Patricia L. Musolino, Allan M. Goldstein
Xin-yang Li, Xue-qi Xie, Bao-chao Wei, Xiao-zheng Sun, Min-xin Chen, Ru-fei Liu, Qing-xu Tao, Yi-heng Huang, Qian Wang, Shuang-shuang Ma, Ling Wei, Rong Xiao, Zhao-yun Liu, Jin-ming Yu, Meng Wu, Dawei ChenXin-yang Li, Xue-qi Xie, Bao-chao Wei, Xiao-zheng Sun, Min-xin Chen, Ru-fei Liu, Qing-xu Tao, Yi-heng Huang, Qian Wang, Shuang-shuang Ma, Ling Wei, Rong Xiao, Zhao-yun Liu, Jin-ming Yu, Meng Wu, Dawei Chen×Abstract
Radiotherapy is a critical modality in cancer treatment, not only to eradicate cancer cells but also to trigger antitumor immunity. IL-21, an immunomodulatory cytokine with potential in cancer therapy, has unexplored synergy with radiotherapy. Our study, leveraging human cancer databases and tissue microarrays, identified a positive correlation between IL-21 and radiotherapy outcomes, particularly in tumor microenvironment (TME) activation. In mouse tumor models, IL-21 combined with radiation significantly enhanced the TME, boosting CD8+ T cell activation and function, reducing tumor burden, and extending survival. Single-cell transcriptome sequencing revealed that the combination of IL-21 and radiation increased the cytotoxicity of effector and memory CD8+ T cells and prevented their exhaustion. These effects were further validated in humanized mice, where IL-21 combined with radiation reduced A549 tumor growth and enhanced CD8+ T cell function. Post-neoadjuvant radiotherapy samples from patients with esophageal cancer showed a positive correlation between IL-21 levels and CD8+ T cell infiltration. Our findings suggest that IL-21 is a promising adjuvant to radiotherapy, potentially improving treatment efficacy through TME enhancement. This study provides a foundation for future clinical exploration of IL-21 for enhancing radiotherapy.
Authors
Xin-yang Li, Xue-qi Xie, Bao-chao Wei, Xiao-zheng Sun, Min-xin Chen, Ru-fei Liu, Qing-xu Tao, Yi-heng Huang, Qian Wang, Shuang-shuang Ma, Ling Wei, Rong Xiao, Zhao-yun Liu, Jin-ming Yu, Meng Wu, Dawei Chen
Chao Li, Pengfei Zhang, Kai Zhang, Jane A. Cook, Weidan Song, Megan Virostek, Lily A. Slotabec, Nadiyeh Rouhi, Mohammed Hazari, Michael I. Adenawoola, Xiaofei Liu, Hao Zhang, Guangyu Zhang, Erica L. Niewold, Qinfeng Li, Yong Fang, Waleed M. Elhelaly, Xue-Nan Sun, Xuejiang Guo, Andrew Lemoff, Yingfeng Deng, Thomas G. Gillette, Ji Li, Philipp E. Scherer, Zhao V. WangChao Li, Pengfei Zhang, Kai Zhang, Jane A. Cook, Weidan Song, Megan Virostek, Lily A. Slotabec, Nadiyeh Rouhi, Mohammed Hazari, Michael I. Adenawoola, Xiaofei Liu, Hao Zhang, Guangyu Zhang, Erica L. Niewold, Qinfeng Li, Yong Fang, Waleed M. Elhelaly, Xue-Nan Sun, Xuejiang Guo, Andrew Lemoff, Yingfeng Deng, Thomas G. Gillette, Ji Li, Philipp E. Scherer, Zhao V. Wang×Abstract
Cardiomyocyte growth is tightly controlled by multiple signaling pathways. Identification of master kinases in this process is essential in exploring potential targets for the treatment of pathological cardiac hypertrophy and heart failure. Here we identified the mTOR-independent activation of ribosomal protein S6 kinase b1 (Rps6kb1) during cardiomyocyte growth. By utilizing phosphoproteomics in primary neonatal rat ventricular myocytes, we revealed Rps6kb1 as one of most activated kinases under growth stimulation. We further demonstrated the role of Rps6kb1 phosphorylation in pathological cardiac hypertrophy and heart failure. We showed that the phosphorylation of multiple sites in Rps6kb1, including T367 in the kinase domain and S418/T421/S424 in the C-terminal domain, is not directly regulated by the activity of mTOR but coupled with the activation of the MEK1/ERK axis. In mice, cardiomyocyte-specific deletion of Rps6kb1 significantly inhibited both constitutively active ERK– and pressure overload–induced cardiac hypertrophy. In contrast, cardiomyocyte-specific overexpression of wild-type Rps6kb1, rather than the phosphorylation-defective mutant, elevated cardiac hypertrophy and augmented pressure overload–induced heart failure. In conclusion, our findings reveal that the MEK/ERK axis primes Rps6kb1 activation through phosphorylation of 2 separate domains of Rps6kb1, which may play an essential role in cardiac hypertrophy and heart failure under hemodynamic stress.
Authors
Chao Li, Pengfei Zhang, Kai Zhang, Jane A. Cook, Weidan Song, Megan Virostek, Lily A. Slotabec, Nadiyeh Rouhi, Mohammed Hazari, Michael I. Adenawoola, Xiaofei Liu, Hao Zhang, Guangyu Zhang, Erica L. Niewold, Qinfeng Li, Yong Fang, Waleed M. Elhelaly, Xue-Nan Sun, Xuejiang Guo, Andrew Lemoff, Yingfeng Deng, Thomas G. Gillette, Ji Li, Philipp E. Scherer, Zhao V. Wang
Mehri Monavarian, Resha Rajkarnikar, Emily Faith Page, Asha Kumari, Liz Quintero Macias, Felipe Massicano, Nam Y. Lee, Sarthak Sahoo, Nadine Hempel, Mohit Kumar Jolly, Lara Ianov, Elizabeth Worthey, Abhyudai Singh, Igor B. Roninson, Eugenia V. Broude, Mengqian Chen, Karthikeyan MythreyeMehri Monavarian, Resha Rajkarnikar, Emily Faith Page, Asha Kumari, Liz Quintero Macias, Felipe Massicano, Nam Y. Lee, Sarthak Sahoo, Nadine Hempel, Mohit Kumar Jolly, Lara Ianov, Elizabeth Worthey, Abhyudai Singh, Igor B. Roninson, Eugenia V. Broude, Mengqian Chen, Karthikeyan Mythreye×Abstract
Anoikis resistance, or evasion of cell death triggered by matrix detachment, is a hallmark of cancer cell survival and metastasis. We showed that repeated exposure to suspension stress followed by recovery under attached conditions leads to development of anoikis resistance. The acquisition of anoikis resistance was associated with enhanced invasion, chemoresistance, and immune evasion in vitro and distant metastasis in vivo. This acquired anoikis resistance was not genetic, persisting for a finite duration without detachment stress, but was sensitive to CDK8/19 mediator kinase inhibition that could also reverse anoikis resistance. Transcriptomic analysis revealed that CDK8/19 kinase inhibition induces bidirectional transcriptional changes in both sensitive and resistant cells, disrupting the balanced reprogramming required for anoikis adaptation and resistance by reversing some resistance-associated pathways and enhancing others. Both anoikis resistance and in vivo metastatic growth of ovarian cancers are sensitive to CDK8/19 inhibition, thereby providing a therapeutic opportunity to both prevent and suppress ovarian cancer metastasis.
Authors
Mehri Monavarian, Resha Rajkarnikar, Emily Faith Page, Asha Kumari, Liz Quintero Macias, Felipe Massicano, Nam Y. Lee, Sarthak Sahoo, Nadine Hempel, Mohit Kumar Jolly, Lara Ianov, Elizabeth Worthey, Abhyudai Singh, Igor B. Roninson, Eugenia V. Broude, Mengqian Chen, Karthikeyan Mythreye
Juan Amaral, Irina Bunea, Arvydas Maminishkis, Maria M. Campos, Francesca Barone, Rohan Gupta, Mitra Farnoodian, Jonathan Newport, M. Joseph Phillips, Ruchi Sharma, David M. Gamm, Kapil Bharti, Richard J. BlanchJuan Amaral, Irina Bunea, Arvydas Maminishkis, Maria M. Campos, Francesca Barone, Rohan Gupta, Mitra Farnoodian, Jonathan Newport, M. Joseph Phillips, Ruchi Sharma, David M. Gamm, Kapil Bharti, Richard J. Blanch×Abstract
Commotio retinae (CR) resulting from retinal trauma can lead to focal photoreceptor degeneration and permanent vision loss. Currently no therapies exist for CR-induced retinal degeneration, in part because of the lack of a large-animal model that replicates human injury pathology and allows testing of therapeutics. Severe CR is clinically characterized by subretinal fluid and focal photoreceptor outer nuclear layer thinning. To develop a porcine CR model, we developed a laser-guided projectile apparatus and optimized projectile delivery procedure using porcine cadaveric eyes embedded in a 3D-printed porcine skull. Scleral and corneal impacts resulted in retinal damage consistent with patient injury, but corneal impacts also led to cornea damage and opacification, which precluded follow-up imaging. In live porcine eyes, scleral impacts of 39.5 m/s induced transient blood-retinal barrier breakdown evidenced by subretinal fluid on optical coherence tomography (OCT), leakage observed on fluorescein and indocyanine green angiography, and transient photoreceptor outer segment disruption seen by OCT and multifocal electroretinography. Impacts above 39.5 m/s induced longer-lasting photoreceptor degeneration but only transient blood-retinal barrier breakdown. This porcine model, combined with clinically relevant imaging and diagnostic modalities, will be valuable for testing the safety and efficacy of therapies to restore vision after focal photoreceptor degeneration.
Authors
Juan Amaral, Irina Bunea, Arvydas Maminishkis, Maria M. Campos, Francesca Barone, Rohan Gupta, Mitra Farnoodian, Jonathan Newport, M. Joseph Phillips, Ruchi Sharma, David M. Gamm, Kapil Bharti, Richard J. Blanch
Ikuhisa Takimoto, Masahiro Shiokawa, Yoshihiro Nishikawa, Takeshi Kuwada, Sakiko Ota, Darryl Joy C. Juntila, Takafumi Yanaidani, Kenji Sawada, Ayako Hirata, Muneji Yasuda, Koki Chikugo, Risa Nakanishi, Masataka Yokode, Yuya Muramoto, Shimpei Matsumoto, Tomoaki Matsumori, Tsutomu Chiba, Hiroshi SenoIkuhisa Takimoto, Masahiro Shiokawa, Yoshihiro Nishikawa, Takeshi Kuwada, Sakiko Ota, Darryl Joy C. Juntila, Takafumi Yanaidani, Kenji Sawada, Ayako Hirata, Muneji Yasuda, Koki Chikugo, Risa Nakanishi, Masataka Yokode, Yuya Muramoto, Shimpei Matsumoto, Tomoaki Matsumori, Tsutomu Chiba, Hiroshi Seno×Abstract
Ulcerative colitis (UC) is a chronic inflammatory condition of the colon that primarily affects the mucosal layer. Previously, we identified autoantibodies against integrin αvβ6 in patients with UC. In this study, we established monoclonal antibodies (mAbs) from patients with UC to reveal the features and functions of these anti–integrin αvβ6 autoantibodies. We identified two shared heavy chain complementarity-determining region 3 (CDR3) amino acid sequences among different patients with UC. Notably, several mAbs contained the RGD sequence in their heavy chain CDR3 that mimicked the key recognition sequence of integrin αvβ6 ligands such as fibronectin. Almost all mAbs selectively reacted with integrin αvβ6 in the presence of divalent cations (Ca2+ and Mg2+) and blocked fibronectin–integrin αvβ6 binding. MAbs that shared the same heavy chain CDR3 amino acid sequence showed differences in reactivity to integrin αvβ6, indicating that the reactivity of these mAbs is also affected by the light chain. Some of the mAbs showed varying degrees of cross-reactivity with integrin αvβ3. The identification of shared CDR3 amino acid sequences in anti–integrin αvβ6 antibodies from several patients with UC suggests a common mechanism underlying their production, which may help elucidate the pathogenesis of UC.
Authors
Ikuhisa Takimoto, Masahiro Shiokawa, Yoshihiro Nishikawa, Takeshi Kuwada, Sakiko Ota, Darryl Joy C. Juntila, Takafumi Yanaidani, Kenji Sawada, Ayako Hirata, Muneji Yasuda, Koki Chikugo, Risa Nakanishi, Masataka Yokode, Yuya Muramoto, Shimpei Matsumoto, Tomoaki Matsumori, Tsutomu Chiba, Hiroshi Seno
Sydney Fobare, Chia Sharpe, Kate Quinn, Kinsey Bryant, Linde A. Miles, Robert L. Bowman, Carolyn Cheney, Casie Furby, Marissa Long, Kaytlynn Fyock, Ben Wronowski, James R. Lerma, Krzysztof Mrózek, Deedra Nicolet, Thomas M. Sesterhenn, Megan E. Johnstone, Jianmin Pan, Shesh N. Rai, Chandrashekhar Pasare, Nives Zimmermann, Wen-Mei Yu, Cheng-Kui Qu, Andrew Carroll, Richard Stone, Eunice S. Wang, Jonathan Kolitz, Bayard Powell, John P. Perentesis, Ann-Kathrin Eisfeld, Erin Hertlein, John C. ByrdSydney Fobare, Chia Sharpe, Kate Quinn, Kinsey Bryant, Linde A. Miles, Robert L. Bowman, Carolyn Cheney, Casie Furby, Marissa Long, Kaytlynn Fyock, Ben Wronowski, James R. Lerma, Krzysztof Mrózek, Deedra Nicolet, Thomas M. Sesterhenn, Megan E. Johnstone, Jianmin Pan, Shesh N. Rai, Chandrashekhar Pasare, Nives Zimmermann, Wen-Mei Yu, Cheng-Kui Qu, Andrew Carroll, Richard Stone, Eunice S. Wang, Jonathan Kolitz, Bayard Powell, John P. Perentesis, Ann-Kathrin Eisfeld, Erin Hertlein, John C. Byrd×Abstract
Mutations in protein tyrosine phosphatase non-receptor type 11 (PTPN11) have been considered late acquired mutations in acute myeloid leukemia (AML) development. Using single-cell DNA sequencing, we found that PTPN11 mutations can occur as initiating events in some patients with AML when accompanied by strong oncogenic drivers, commonly NPM1 mutations. The resulting AML has a diverse set of variably differentiated myeloid cells with few myeloid cells that lack leukemic mutations. The role of Ptpn11 as a codriver was confirmed in a murine model that exhibits an AML phenotype with a comparable immune diversity that is serially engraftable and reconstituted from early precursor cells. Furthermore, lineage-negative bone marrow cells from these mice reconstitute the full diversity of mature myeloid cells, and these cells exhibit an altered cytokine response after physiologic stimulation. Our work highlights how PTPN11-mutated AML is derived from a multitude of codominant and late acquired aberrations that have a previously unrecognized differentiated myeloid clonal expansion potentially contributing to pathogenesis of the disease.
Authors
Sydney Fobare, Chia Sharpe, Kate Quinn, Kinsey Bryant, Linde A. Miles, Robert L. Bowman, Carolyn Cheney, Casie Furby, Marissa Long, Kaytlynn Fyock, Ben Wronowski, James R. Lerma, Krzysztof Mrózek, Deedra Nicolet, Thomas M. Sesterhenn, Megan E. Johnstone, Jianmin Pan, Shesh N. Rai, Chandrashekhar Pasare, Nives Zimmermann, Wen-Mei Yu, Cheng-Kui Qu, Andrew Carroll, Richard Stone, Eunice S. Wang, Jonathan Kolitz, Bayard Powell, John P. Perentesis, Ann-Kathrin Eisfeld, Erin Hertlein, John C. Byrd
Xunde Wang, Meghann Smith, Sayuri Kamimura, Quan Li, Niharika Shah, Martha Quezado, Luis E.F. Almeida, Sebastian Vogel, Mickias B. Tegegn, Kevin Y. Sun, Rafael Villasmil, Chengyu Liu, William A. Eaton, Swee Lay Thein, Zenaide M.N. QuezadoXunde Wang, Meghann Smith, Sayuri Kamimura, Quan Li, Niharika Shah, Martha Quezado, Luis E.F. Almeida, Sebastian Vogel, Mickias B. Tegegn, Kevin Y. Sun, Rafael Villasmil, Chengyu Liu, William A. Eaton, Swee Lay Thein, Zenaide M.N. Quezado×Abstract
Growing evidence indicates that PKLR, the gene for pyruvate kinase (PK), is a genetic modifier of the sickle cell phenotype. Coinheritance of specific PKLR variants is associated with increased pain-related hospitalization and can trigger sickle cell disease (SCD) phenotypes in asymptomatic carriers. PK deficiency disrupts RBC glycolysis, leading to ATP deficits and accumulation of 2,3-diphosphoglycerate, which exacerbates sickling in SCD. Using CRISPR-Cas9, we generated null mutations in Pklr [Pklr(13ntdel/13ntdel) or Pklr(246ntdel/246ntdel)] specific for the RBC isoform (PKR) in Townes mice that were homozygous (SS) or heterozygous (AS) for the human sickle globin gene, or homozygous for human hemoglobin A (AA, controls), to investigate the effect of PKR deficiency on the sickle phenotype in mice. PKR-deficient AA and AS mice developed severe anemia, reticulocytosis, and substantial spleen and liver iron deposits. Unlike what is observed in humans, PKR deficiency in AS and SS mice surprisingly decreased sickling, but it was also associated with increased extramedullary hematopoiesis and mitochondrial retention in mature RBCs. These results demonstrate the differential effect of Pklr mutations on the phenotype of both AS and SS mouse models, offering insights into the complex role of PKR deficiency in SCD pathology.
Authors
Xunde Wang, Meghann Smith, Sayuri Kamimura, Quan Li, Niharika Shah, Martha Quezado, Luis E.F. Almeida, Sebastian Vogel, Mickias B. Tegegn, Kevin Y. Sun, Rafael Villasmil, Chengyu Liu, William A. Eaton, Swee Lay Thein, Zenaide M.N. Quezado
×Abstract
In this study, we used single-cell RNA sequencing to delineate the contributions of muscle-resident Schwann cells to neuromuscular junction (NMJ) remodeling by comparing a model of stable innervation with models of reinnervation following partial or complete denervation. We discovered multiple distinct Schwann cell subtypes, including a terminal Schwann cell subtype integral to the denervation-reinnervation cycle, identified by a transcriptomic signature indicative of cell migration and polarization. The data also characterize 3 myelin Schwann cell subtypes, which are distinguished based on enrichment of genes associated with myelin production, mesenchymal differentiation, or collagen synthesis. Importantly, SPP1 signaling emerged as a pivotal regulator of NMJ dynamics, promoting Schwann cell proliferation and muscle reinnervation across nerve injury models. These findings advance our understanding of NMJ maintenance and regeneration and underscore the therapeutic potential of targeting specific molecular pathways to treat neuromuscular and neurodegenerative disorders.
Authors
Steve D. Guzman, Ahmad Abu-Mahfouz, Carol S. Davis, Lloyd P. Ruiz, Peter C.D. Macpherson, Susan V. Brooks
×Abstract
The skin lesion erythema migrans (EM) is the first clinical sign of Lyme disease, an infection due to the tick-transmitted bacterium Borrelia burgdorferi (Bb). Previously, we used scRNA-Seq to characterize the cutaneous immune response in the EM lesion, focusing on B cells. Here, with an expanded sample size, we profiled T cell responses in EM lesions compared to autologous uninvolved skin. In addition to CD4+ T cell subsets known to be abundant in the EM lesion, we identified clonally expanded CD8+GZMK+IFNG+ T cells that comprised cells with high or intermediate IFNG expression. These cells exhibited significant differential expression of IFN-regulated genes and included subsets with low cytotoxic gene expression, suggesting an inflammatory potential that may contribute to early defense against Bb within the EM lesion. In addition, we found that endothelial cells, fibroblasts, and pericytes were the main producers of key T cell–recruiting chemokines. These studies using single-cell transcriptomics with adaptive immune receptor sequencing provide a comprehensive interrogation of the cutaneous T cell response to Bb infection and insight into the orchestration of the skin barrier defense to this vector-borne pathogen.
Authors
Edel Aron, Hailong Meng, Alexia A. Belperron, Paraskevas Filippidis, Kenneth R. Dardick, Steven H. Kleinstein, Linda K. Bockenstedt
Rene Weyer, Katharina Völker, Tamara Potapenko, Lisa Krebes, Marco Abeßer, Anna-Lena Friedrich, Eva Lessmann, Ali Khadim, Clemens Ruppert, Elie El Agha, Dalia Sheta, Andreas Beilhack, Daniel V. Santi, Eric L. Schneider, Michaela Kuhn, Swati DabralRene Weyer, Katharina Völker, Tamara Potapenko, Lisa Krebes, Marco Abeßer, Anna-Lena Friedrich, Eva Lessmann, Ali Khadim, Clemens Ruppert, Elie El Agha, Dalia Sheta, Andreas Beilhack, Daniel V. Santi, Eric L. Schneider, Michaela Kuhn, Swati Dabral×Abstract
Activation of lung fibroblasts in response to epithelial injury and inflammation provokes pulmonary fibrosis (PF). Endogenous molecular brakes counteracting fibroblast activity can be targets for therapies. Preclinical studies of synthetic C-type natriuretic peptide (CNP) indicated that this hormone might provide such a brake. As shown here, CNP exerts antifibrotic effects in cultured lung fibroblasts as well as in precision cut lung slices from patients with PF, supporting clinical relevance. Therefore, augmenting or supplementing endogenous CNP could improve the treatment of such patients. To unravel whether paracrine CNP counteracts inflammation-driven PF, we studied mice with fibroblast-restricted KO of guanylyl-cyclase-B (GC-B), its cGMP-synthesizing receptor. Fibroblast GC-B-KO mice had enhanced bleomycin-induced lung inflammation, with increased expression of proinflammatory, profibrotic cytokines. Nevertheless, subsequent PF was not exacerbated. Molecular studies revealed that inflammation led to inhibition of CNP signaling in resident myofibroblasts, namely GC-B downregulation and induction of CNP/cGMP-degrading pathways. Despite this, a single s.c. injection of the recently developed long-acting CNP analog, MS~[Gln6,14]CNP-38, abrogated experimental lung inflammation and fibrosis. We conclude that CNP signaling in lung fibroblasts has antiinflammatory and antifibrotic effects. Attenuation of this endogenous brake participates in the pathogenesis of PF, and rescuing this pathway with long-acting CNP-analogs may have therapeutic potential.
Authors
Rene Weyer, Katharina Völker, Tamara Potapenko, Lisa Krebes, Marco Abeßer, Anna-Lena Friedrich, Eva Lessmann, Ali Khadim, Clemens Ruppert, Elie El Agha, Dalia Sheta, Andreas Beilhack, Daniel V. Santi, Eric L. Schneider, Michaela Kuhn, Swati Dabral
Liu Di, Jiang-bo Fan, Rui Wang, You Li, Wan-da Bi, Si-yuan Huang, Heng-hai Nie, Xi-feng Feng, Hua-cai Zhang, Juan Du, Xiao-fei Huang, An-yong Yu, Zhe Xu, Fei Xia, Jian-xin Jiang, Shuang-shuang Dai, Xiang Xu, Zhen Wang, Ling ZengLiu Di, Jiang-bo Fan, Rui Wang, You Li, Wan-da Bi, Si-yuan Huang, Heng-hai Nie, Xi-feng Feng, Hua-cai Zhang, Juan Du, Xiao-fei Huang, An-yong Yu, Zhe Xu, Fei Xia, Jian-xin Jiang, Shuang-shuang Dai, Xiang Xu, Zhen Wang, Ling Zeng×Abstract
Sepsis, a systemic inflammatory response to infection, remains a leading cause of mortality in intensive care units, with sepsis-induced immunosuppression being a critical pathophysiological process. In this study, we investigated the role of histone deacetylase 1 (HDAC1) in sepsis-induced CD8+ T cell exhaustion, a key driver of immunosuppression. Clinical analyses of patients with sepsis revealed that reduced peripheral blood lymphocyte levels, particularly CD8+ T cell depletion, strongly correlated with worsened outcomes. In a murine sepsis model, single-cell RNA-Seq revealed a significant decrease in the proportion of CD8+ T cells and an increase in the proportion of exhausted CD8+ T cells in mouse lungs. Adoptive transfer of CD8+ T cells effectively reduced sepsis mortality by preserving organ function. We further demonstrated that HDAC1 expression was significantly upregulated in CD8+ T cells from patients with sepsis. In vitro studies showed that HDAC1 inhibition preserved CD8+ T cell function by maintaining T cell activity and reducing the expression of inhibitory molecules such as PD-1. Pharmacological inhibition of HDAC1 reduced mortality and reversed CD8+ T cell exhaustion by restoring the balance between activator protein-1 (AP-1) and nuclear factor of activated T cells (NFAT). Additionally, we found that HDAC1 directly interacted with NFAT1, promoting its nuclear translocation and further enhancing the expression of inhibitory molecules. Our findings highlight HDAC1 as a potential therapeutic target for sepsis-induced immunosuppression. By elucidating the molecular mechanisms underlying HDAC1-mediated immunosuppression, we have provided potential strategies for developing immunomodulatory therapies for the treatment of sepsis.
Authors
Liu Di, Jiang-bo Fan, Rui Wang, You Li, Wan-da Bi, Si-yuan Huang, Heng-hai Nie, Xi-feng Feng, Hua-cai Zhang, Juan Du, Xiao-fei Huang, An-yong Yu, Zhe Xu, Fei Xia, Jian-xin Jiang, Shuang-shuang Dai, Xiang Xu, Zhen Wang, Ling Zeng
Natalia A. Kuzmina, Sivakumar Periasamy, Kritika Kedarinath, Keziah Hernandez, Caroline Atyeo, S. Moses Dennison, Kan Li, Daniel Bedinger, Sharon L. Schendel, Georgia D. Tomaras, Hanif Ali, Galit Alter, Erica Ollmann Saphire, Alexander BukreyevNatalia A. Kuzmina, Sivakumar Periasamy, Kritika Kedarinath, Keziah Hernandez, Caroline Atyeo, S. Moses Dennison, Kan Li, Daniel Bedinger, Sharon L. Schendel, Georgia D. Tomaras, Hanif Ali, Galit Alter, Erica Ollmann Saphire, Alexander Bukreyev×Abstract
Antibody-dependent enhancement (ADE) of infection is a well-described phenomenon for several viruses, including dengue, Ebola, respiratory syncytial virus, and HIV. ADE occurs when virus-antibody complexes engage Fc receptors (FcRs) and virus-specific receptors, enhancing infection under conditions of incomplete neutralization. The Coronavirus Immunotherapeutic Consortium (CoVIC) assembled a comprehensive dataset of functional properties for over 400 mAbs, enabling direct comparison of neutralization, Fc-mediated functions, receptor binding, and infection of immune cells. Infection rates in most primary human immune cell types were low, with modest increases observed for some mAbs. In contrast, macrophages were more susceptible to SARS-CoV-2 and exhibited substantial ADE with select mAbs. ADE was completely inhibited by FcR blockade and significantly reduced by antibody- or ceftazidime-mediated blocking of angiotensin-converting enzyme 2 (ACE2). Neutralization potency did not correlate with ADE, as both strongly and weakly neutralizing antibodies induced enhancement. Instead, ADE magnitude depended on an antibody’s ability to block spike protein binding to ACE2. Importantly, ADE resulted in productive infection with release of infectious virus. Evaluation of antibodies against the BA.1 (Omicron) variant revealed reduced or lost ADE for most mAbs, with increased ADE observed for several mAbs relative to the USA-WA1/2020 strain.
Authors
Natalia A. Kuzmina, Sivakumar Periasamy, Kritika Kedarinath, Keziah Hernandez, Caroline Atyeo, S. Moses Dennison, Kan Li, Daniel Bedinger, Sharon L. Schendel, Georgia D. Tomaras, Hanif Ali, Galit Alter, Erica Ollmann Saphire, Alexander Bukreyev
Romaila Abd-El-Raouf, Jakob Harrison-Gleason, Jinhee Kim, Ching Man Wai, Kayla L. Yerlioglu, Catarina Ananias-Saez, Alec Ksiazek, Jeffrey T. Poomkudy, Mariluz Araínga, Deepanwita Bose, Claudia Cicala, James Arthos, Francois J. Villinger, Ramon Lorenzo-Redondo, Elena MartinelliRomaila Abd-El-Raouf, Jakob Harrison-Gleason, Jinhee Kim, Ching Man Wai, Kayla L. Yerlioglu, Catarina Ananias-Saez, Alec Ksiazek, Jeffrey T. Poomkudy, Mariluz Araínga, Deepanwita Bose, Claudia Cicala, James Arthos, Francois J. Villinger, Ramon Lorenzo-Redondo, Elena Martinelli×Abstract
We previously demonstrated that blocking TGF-β with galunisertib, a safe, orally available small drug, reactivated latent SIV in vivo by shifting T cells toward a transitional effector phenotype. Here, we investigated the mechanisms underlying this effect using single-cell RNA sequencing, metabolic profiling, and high-dimensional spectral flow cytometry of samples from SIV-infected, antiretroviral therapy–treated (ART-treated) macaques before and after galunisertib. To characterize virus-transcribing, infected cells during ART, we developed a novel, sensitive SIV Transcripts Capture Assay (SCAP) that detected 127 SIV-expressing cells within lymph node single-cell transcriptome libraries. Galunisertib drove broad metabolic reprogramming in CD4+ T cells, with transcriptional upregulation of inflammatory and mitochondrial biosynthesis pathways, confirmed by Seahorse profiling. Metabolomics revealed increased energy metabolites and amino acids and enhanced metabolic flux without proliferation. SIV transcript–positive cells before galunisertib were metabolically quiescent compared with cells without detectable viral transcripts. After galunisertib, virus-expressing cells showed a dramatic metabolic activation, with upregulation of glycolysis, fatty acid metabolism, and TNF-α signaling. High-dimensional flow cytometry demonstrated effects beyond CD4+ T cells, including fewer tissue-resident memory T cells, but more inflammatory macrophages. In conclusion, SCAP represents a specific tool for characterizing rare SIV-infected cells transcribing virus during ART, and it reveals TGF-β as a key mediator of viral latency in vivo through metabolic suppression.
Authors
Romaila Abd-El-Raouf, Jakob Harrison-Gleason, Jinhee Kim, Ching Man Wai, Kayla L. Yerlioglu, Catarina Ananias-Saez, Alec Ksiazek, Jeffrey T. Poomkudy, Mariluz Araínga, Deepanwita Bose, Claudia Cicala, James Arthos, Francois J. Villinger, Ramon Lorenzo-Redondo, Elena Martinelli
Giray Eryilmaz, Yilmaz Yucehan Yazici, Radu Marches, Eleni P. Mimitou, Lisa Kenyon-Pesce, Kim Handrejk, Sonia Jangra, Michael Schotsaert, Adolfo García-Sastre, George A. Kuchel, Jacques Banchereau, Duygu UcarGiray Eryilmaz, Yilmaz Yucehan Yazici, Radu Marches, Eleni P. Mimitou, Lisa Kenyon-Pesce, Kim Handrejk, Sonia Jangra, Michael Schotsaert, Adolfo García-Sastre, George A. Kuchel, Jacques Banchereau, Duygu Ucar×Abstract
BACKGROUND The SARS-CoV-2 pandemic provided a rare opportunity to study how human immune responses develop to a novel viral antigen delivered through different vaccine platforms. However, to date, no study has directly compared immune responses to all 3 FDA-approved COVID-19 vaccines at single-cell multiomic resolution.METHODS We longitudinally profiled SARS-CoV-2–naive adults (n = 31) vaccinated with BNT162b2, mRNA-1273, or Ad26.COV2.S, integrating plasma cytokines, antibody titers, and single-cell multiomic data (DOGMA-Seq).RESULTS We discovered a distinct, transient IFN program termed ISG-dim, which emerged specifically 1–2 days after the first mRNA dose in approximately 10% of myeloid cells. This state was characterized by ISGF3 complex activation and its target genes (e.g., MX1, MX2, DDX58), with transcriptional and epigenetic profiles distinct from the robust IFN program observed after mRNA boosting or a single Ad26.COV2.S dose (ISG-high). In vitro stimulation of human monocytes showed that IFN-α alone recapitulates ISG-dim, whereas both IFN-α and IFN-γ are required for ISG-high.CONCLUSION These findings define dose-dependent IFN programming in human myeloid cells and highlight mechanistic differences between priming and boosting, with implications for optimizing vaccine platform choice, dose scheduling, and formulation.FUNDING NIH grants AI142086, U19 AI135972, U01 AI165452, U01 AI165452, R01 AI160706, and P30 AG067988.
Authors
Giray Eryilmaz, Yilmaz Yucehan Yazici, Radu Marches, Eleni P. Mimitou, Lisa Kenyon-Pesce, Kim Handrejk, Sonia Jangra, Michael Schotsaert, Adolfo García-Sastre, George A. Kuchel, Jacques Banchereau, Duygu Ucar
Collin R. Diedrich, Tara Rutledge, Janelle L. Gleim, Christopher Kline, Pauline Maiello, Jessica M. Medrano, H. Jacob Borish, Harris B. Chishti, Justin L. Gaines, Edwin Klein, Forrest Hopkins, Jacob E. Klein, Daniel Fillmore, Kara Kracinovsky, Jaime Tomko, Jennifer Schober, Sarah M. Fortune, Michael C. Chao, JoAnne L. Flynn, Zandrea Ambrose, Philana Ling LinCollin R. Diedrich, Tara Rutledge, Janelle L. Gleim, Christopher Kline, Pauline Maiello, Jessica M. Medrano, H. Jacob Borish, Harris B. Chishti, Justin L. Gaines, Edwin Klein, Forrest Hopkins, Jacob E. Klein, Daniel Fillmore, Kara Kracinovsky, Jaime Tomko, Jennifer Schober, Sarah M. Fortune, Michael C. Chao, JoAnne L. Flynn, Zandrea Ambrose, Philana Ling Lin×Abstract
Coinfection with both HIV and M. tuberculosis (Mtb) results in disseminated tuberculosis (TB) and accelerated HIV progression. Despite greater access to antiretroviral treatment (ART), it remains unclear whether suppression of HIV replication protects against severe Mtb infection. Here, using a macaque model of SIV/Mtb coinfection, we investigated whether treatment of SIV infection with ART influenced control of a subsequent Mtb challenge compared with SIV-infected macaques that were not treated with ART. Macaques were first infected with SIVB670, SIVB670 with ART, or saline followed by a low-dose Mtb inoculation with serial clinical and PET-CT imaging assessments. At necropsy, gross pathology, viremia, bacterial burden, and immunologic parameters were compared. SIV-TB animals had greater gross pathology and total bacterial burden than TB-only and SIV/ART/TB groups. However, despite normal blood CD4 counts and undetectable SIV RNA, SIV/ART/TB macaques showed similar clinical parameters and extrapulmonary involvement as SIV/TB animals. Analysis of barcoded-Mtb suggests that ART control of SIV replication did not prevent Mtb extrapulmonary dissemination. These data indicate that people living with HIV on ART remain at high risk of bacterial dissemination and extrapulmonary TB disease. Understanding the mechanisms of extrapulmonary spread and disease severity during HIV/TB coinfection remains an important issue.
Authors
Collin R. Diedrich, Tara Rutledge, Janelle L. Gleim, Christopher Kline, Pauline Maiello, Jessica M. Medrano, H. Jacob Borish, Harris B. Chishti, Justin L. Gaines, Edwin Klein, Forrest Hopkins, Jacob E. Klein, Daniel Fillmore, Kara Kracinovsky, Jaime Tomko, Jennifer Schober, Sarah M. Fortune, Michael C. Chao, JoAnne L. Flynn, Zandrea Ambrose, Philana Ling Lin
Martin Hindermann, Justus B.H. Wilke, Yasmina Curto, Stefan N. Oline, Vinicius Daguano Gastaldi, Umer Javed Butt, Rakshit Dadarwal, Umut Çakır, Anja Ronnenberg, Kurt Hammerschmidt, Susann Boretius, Anastassia Stoykova, Anton B. Tonchev, Klaus-Armin Nave, Manvendra Singh, Hannelore EhrenreichMartin Hindermann, Justus B.H. Wilke, Yasmina Curto, Stefan N. Oline, Vinicius Daguano Gastaldi, Umer Javed Butt, Rakshit Dadarwal, Umut Çakır, Anja Ronnenberg, Kurt Hammerschmidt, Susann Boretius, Anastassia Stoykova, Anton B. Tonchev, Klaus-Armin Nave, Manvendra Singh, Hannelore Ehrenreich×Abstract
Among the known genetic causes of syndromic autism spectrum disorders (ASDs) are transcription factor deficiencies. In this regard, haploinsufficiency of the zinc finger and broad complex, tramtrack, bric and brac domain–containing protein 20 (ZBTB20) leads to a prototypical clinical picture, referred to as Primrose syndrome, comprising severe ASD symptoms together with intellectual disability. Here, we present a comprehensive behavioral and phenotypical characterization of Zbtb20+/– mice, a construct valid model of this thus far untreatable human condition. Zbtb20+/– mice exhibited diminished sociability, reduced vocalization, distinct repetitive behaviors, impaired cognitive flexibility, hyperactivity, and hypoalgesia. Magnetic resonance imaging revealed increased volumes of hippocampus, cerebellum, brain matter, and whole brain, confirmed by postmortem brain weight measurements. Due to our previous observation of enhanced ZBTB20 expression in CA1 pyramidal neurons upon recombinant human erythropoietin (rhEPO) injections, we anticipated a mitigating effect through rhEPO treatment of Zbtb20 deficiency/Primrose syndrome. Indeed, after 3 weeks of alternate-day rhEPO injections, a remarkable improvement in the behavioral phenotype was observed. Our results highlight rhEPO as promising treatment for Primrose syndrome.
Authors
Martin Hindermann, Justus B.H. Wilke, Yasmina Curto, Stefan N. Oline, Vinicius Daguano Gastaldi, Umer Javed Butt, Rakshit Dadarwal, Umut Çakır, Anja Ronnenberg, Kurt Hammerschmidt, Susann Boretius, Anastassia Stoykova, Anton B. Tonchev, Klaus-Armin Nave, Manvendra Singh, Hannelore Ehrenreich