Dermatology
Abstract
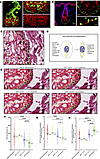
Nicotinamide adenine dinucleotide (NAD⁺) is essential for cellular metabolism, DNA repair, and stress responses. NAD+ is synthesized from nicotinamide, nicotinic acid (collectively termed niacin), and tryptophan. In humans, deficiencies in these nutrients result in pellagra, marked by dermatitis, diarrhea, and dementia. The dermatitis associated with pellagra typically manifests as photodermatosis in sun-exposed areas. This study examined the effects of NAD+ deficiency on skin homeostasis using epidermis-specific Nampt conditional knockout (cKO) mice. These mice displayed substantial NAD⁺ depletion, reduced poly(ADP-ribose) polymerase (PARP) activity, and increased DNA damage. Consequently, Nampt cKO mice developed spontaneous skin inflammation and epidermal hyperplasia. RNA sequencing and immunohistochemical analyses demonstrated increased interleukin-36 (IL-36) cytokine expression, suggesting that DNA repair-related genomic stress triggers keratinocyte-driven IL-36 production, which promotes inflammation. Furthermore, reduced collagen17A1 expression and elevated thymic stromal lymphopoietin (TSLP) levels were observed. NAD+ repletion by transdermal supplementation of nicotinamide mononucleotide (NMN) suppressed the rise of IL-36 levels and skin inflammation. These findings underscore the importance of Nampt-mediated NAD⁺ metabolism for epidermal stability and indicate that NAD⁺ depletion may contribute to IL-36-mediated skin inflammation, offering insights for therapeutic strategies in inflammatory skin disorders.
Authors
Taiki Seki, Jun-Dal Kim, Yasuhito Yahara, Hitoshi Uchida, Keisuke Yaku, Mariam Karim, Teruhiko Makino, Tadamichi Shimizu, Takashi Nakagawa
Abstract
Acne vulgaris is a common skin condition involving complex interactions among lipid-secreting sebaceous glands, keratinocytes, immune cells, and microbiota. While retinoids are effective for treating acne, disease pathogenesis remains poorly understood. In particular, it remains unclear how different subtypes of acne, including inflammatory (pustular) and noninflammatory (comedonal) lesions, vary in gene expression, signaling, and sebaceous gland involvement. Here, we performed spatial transcriptomics on healthy, nonlesional, comedonal, and pustular acne skin using a custom panel targeting sebaceous differentiation, lipid metabolism, and retinoid signaling pathways. We also designed a specialized segmentation pipeline to improve transcript assignment in the spatially complex sebaceous gland. Our analyses identified a PPARG+ transitional basal cell state in sebocytes and revealed that comedonal skin upregulates sebogenesis genes, whereas pustular skin downregulates sebogenesis. Both lesion types exhibited increased AP-1 transcription factors and elevated FABP5, a chaperone that blunts retinoic acid receptor signaling. Finally, we demonstrated that an AP-1 inhibitor, T-5224, downregulates FABP5 in human keratinocytes and reduces pustule formation in a mouse model of high-fat diet–induced folliculitis. Altogether, these findings indicate that altered lipogenesis, retinoid signaling, and keratinocyte differentiation are key features of acne, and nominate AP-1 and FABP5 as potential therapeutic targets.
Authors
Joseph S. Durgin, Natalia A. Veniaminova, Thomas J. Huyge, Shih-Ying Tsai, Jennifer Fox, Yuli Cai, Mrinal K. Sarkar, Lam C. Tsoi, Johann E. Gudjonsson, Sunny Y. Wong
Abstract
Adaptive immune responses are widely considered the primary drivers of chronic inflammation in autoimmune disease, yet increasing evidence suggests that dysregulated myeloid cells play a central role in sustaining tissue damage. Salt-inducible kinases (SIKs) regulate immune cell activation, and their pharmacological inhibition can promote a shift from proinflammatory toward an immunoregulatory phenotype. We investigated whether selective inhibition of SIK2 and SIK3 with GLPG3970 could reprogram monocytes, macrophages, and dendritic cells, and we assessed pharmacological effects on activated T and B cells. Preclinical studies in mouse models of colitis, psoriasis, and arthritis demonstrated that SIK2/SIK3 inhibition reduced inflammatory activity and promoted immunoregulatory and tolerogenic-associated pathways. Clinical signal-detection studies in ulcerative colitis, psoriasis, and rheumatoid arthritis revealed signs of clinical and biological activity in ulcerative colitis and psoriasis. These findings suggest that myeloid cell dysfunction and impaired myeloid phenotype switching contribute to chronic inflammation in autoimmune diseases and that therapeutic targeting of SIK2/SIK3 holds the potential to restore immune balance by converting proinflammatory into regulatory pathways. Collectively, this work supports SIK2/SIK3 inhibition as a potential treatment strategy for myeloid cell–driven chronic inflammatory conditions.
Authors
Steve De Vos, Nicolas Desroy, Susan J. Bellaire, Anna Pereira Fernandes, Stéphanie Lavazais, Didier Merciris, Carole Delachaume, Catherine Robin-Jagerschmidt, Adrien Cosson, Angela Lazaryan, Nancy Van Osselaer, David Amantini, Christophe Peixoto, Maikel L. Colli, Thomas Van Eeckhoutte, Tiina Hakonen, Magali Constant, Alberto Garcia-Hernandez, Rahul Barron, Geert D’Haens, Wulf O. Böcher
Abstract
Chronic hyperglycemia changes the expression of various transcription factors and mRNA transcripts, which impair the cellular functionality and delayed wound healing. ZEB2 (zinc finger E-box binding homeobox 2), a key transcription factor maintains the tissue specific macrophage identities, however, its role in regulating macrophage polarization during wound healing under hyperglycemic conditions remains unclear. Here, we have found that persistent hyperglycemia increases ZEB2 expression in wound macrophages via histone acetylation, contributing to chronic inflammation, and delayed wound healing. Exposure to high glucose levels activates P300/CBP, a transcriptional coactivator involved in histone acetylation, enhances ZEB2 expression in wound macrophages. The forced expression of ZEB2 shifts macrophage polarity toward a pro-inflammatory state by upregulating myeloid lineage directed transcription factors (MLDTFs). Conversely, silencing Zeb2 at the wound site reduced hyperglycemia induced macrophage inflammation. Topical application of C646, an inhibitor of P300, at the wound edges of streptozotocin induced high-fat diet fed diabetic mice significantly decreased ZEB2 expression, reduced inflammation and accelerated wound healing. Therefore, targeted inhibition of P300 represents a promising therapeutic strategy for improving diabetic wound healing by modulating ZEB2 driven inflammation in wound macrophages.
Authors
Soumyajit Roy, Debarun Patra, Palla Ramprasad, Shivam Sharma, Parul Katiyar, Ashvind Bawa, Kanhaiya Singh, Kulbhushan Tikoo, Suman Dasgupta, Chandan K. Sen, Durba Pal
Abstract
Psoriasis is a chronic inflammatory dermatosis characterized by pathological keratinocyte hyperproliferation and dysregulated immune activation. While ubiquitin-specific peptidase 16 (USP16) has been implicated in modulating multiple cellular signaling pathways, its functional role in psoriatic pathogenesis remains poorly understood. Our investigation revealed pronounced upregulation of USP16 expression in psoriatic epidermis compared to normal controls. Keratinocyte-specific USP16 knockdown demonstrated remarkable therapeutic efficacy, significantly ameliorating characteristic psoriatic phenotypes including epidermal hyperplasia and inflammatory infiltration. RNA sequencing analysis showed that USP16 has substantial effects on cell cycle transition and keratinocytes proliferation. Through KEGG analysis, it was found that USP16 primarily regulates the NLRP3 signaling pathway, leading to enhanced cell proliferation and inflammation. Mechanically, USP16 directly binds to the NLRP3 protein to eliminate K48 ubiquitination modification, enhancing the stability of the NLRP3 protein, activating inflammasome activity. Further studies showed that the therapeutic effect of reducing USP16 on psoriasis progression were counteracted by an NLRP3 activator and keratinocyte-specific NLRP3 overexpression adenovirus. Collectively, these results shed light on how USP16 promotes NLRP3 signaling in keratinocytes, exacerbating psoriasis development. This positive regulation highlights the potential of USP16 as a therapeutic target for psoriasis.
Authors
Nan Wang, Fangqian Guan, Yifan Lin, Bohao Sun, Jindan Dai, Xiejun Xu, Weibo Tang, Yanhua Ren, Xuliang Huang, Wenjie Gao, Xixi Chen, Litai Jin, Weitao Cong, Zhongxin Zhu
Abstract
BACKGROUND Icotrokinra is the first and only targeted oral peptide that selectively binds the IL-23 receptor with high affinity to precisely inhibit IL-23 signaling. Icotrokinra demonstrated high rates of complete skin clearance and durable disease control in the phase IIb trial, FRONTIER-1, and its long-term extension, FRONTIER-2, in participants with moderate-to-severe plaque psoriasis. This study evaluated systemic and skin pharmacodynamic response of icotrokinra and its relationship to clinical response in FRONTIER participants.METHODS FRONTIER-1 participants received icotrokinra or placebo for 16 weeks. FRONTIER-2 followed participants for up to 1 year of treatment; placebo participants transitioned to icotrokinra after week 16. Systemic pharmacodynamic changes were assessed in serum through week 52. Skin pharmacodynamic changes were assessed using transcriptomic analysis of skin biopsies and protein quantification in tape-strip samples through week 16.RESULTS Icotrokinra dose-dependently reduced serum levels of the IL-23/IL-17 axis and psoriasis disease biomarkers through week 52, with maximal reductions observed with the highest 100 mg twice-daily dose. Proteomic analyses showed icotrokinra selectively blocked IL-23–driven inflammation without broader impacts on circulating proteins, including serum IL-23 levels. Sixteen weeks of icotrokinra, but not placebo, reduced expression of psoriasis-associated genes in lesional skin. Icotrokinra treatment also reduced psoriasis-relevant proteins in week 16 lesional skin tape-strips to levels comparable to nonlesional samples.CONCLUSION Icotrokinra induced a dose-dependent pharmacodynamic response, with early (week 4) and sustained (week 52) reductions in biomarkers of IL-23 pathway activation and psoriasis disease severity, which correlated with clinical response.TRIAL REGISTRATION ClinicalTrials.gov: NCT05223868, NCT05364554.FUNDING Johnson & Johnson.
Authors
David Strawn, James G. Krueger, Robert Bissonnette, Kilian Eyerich, Laura K. Ferris, Amy S. Paller, Andreas Pinter, Dylan Richards, Elizabeth Y. Chen, Kate Paget, Daniel Horowitz, Roohid Parast, Joshua J. Rusbuldt, Jocelyn Sendecki, Sunita Bhagat, Lynn P. Tomsho, Ching-Heng Chou, Marta E. Polak, Brice E. Keyes, Emily Bozenhardt, Yuan Xiong, Wangda Zhou, Cynthia DeKlotz, Paul Newbold, Dawn M. Waterworth, Megan Miller, Takayuki Ota, Ya-Wen Yang, Monica W.L. Leung, Lloyd S. Miller, Carolyn A. Cuff, Bradford McRae, Darren Ruane, Arun K. Kannan
Abstract
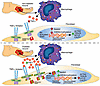
Uncovering the early interactions and spatial distribution of dermal fibroblasts and immune cells in treatment-naïve diffuse cutaneous systemic sclerosis (dcSSc) patients is critical to understanding the earliest events of skin fibrosis. We generated an integrated multiomic dataset of early, treatment-naïve dcSSc skin. Skin biopsies were analyzed by single-nuclei multiome sequencing (snRNA-seq and snATAC-seq) and two different spatial transcriptomic methods to comprehensively determine the molecular changes in these individuals. We identified an immunomodulatory niche within the papillary, hypodermis, and vascular regions that are enriched for activated myeloid cells and fibroblasts characterized by expression of genes such as CXCL12, APOE, and C7. Pathway analyses showed significant enrichment of PI3K-AKT-mTOR signaling pathway expression in these cellular niches, driven by profibrotic growth factor signaling networks. Macrophage subclustering showed SSc-specific macrophage activation of the IL6-JAK-STAT signaling and the enrichment of oxidative phosphorylation pathways. Ligand-receptor analysis revealed that SSc macrophages secrete PDGF and TGF-β to activate the SSc-dominant fibroblast subclusters. Spatial transcriptomic analyses showed monocyte-derived MRC1+ macrophages express PDGF near PDGFRhighTHY1high fibroblasts. Multi-omic data integration and spatial transcriptomic neighborhood analysis revealed the co-localization of fibroblasts, macrophages, and T cells around the vasculature. These data suggest that interactions between activated immune cells and immunomodulatory fibroblasts around vascular niches are an early event in scleroderma pathogenesis.
Authors
Helen C. Jarnagin, Rezvan Parvizi, Zhiyun Gong, Rosemary Gedert, Xianying Xing, Lam (Alex) C. Tsoi, Rachael Bogle, Madeline J. Morrisson, Laurent Perreard, Patricia A. Pioli, Fred Kolling IV, Johann E. Gudjonsson, Dinesh Khanna, Michael L. Whitfield
Abstract
Oral lichen planus (OLP) is a recalcitrant inflammatory disease with potential for malignant transformation, involving a cytotoxic CD8+ T cells-mediated basal keratinocyte apoptosis. However, it lacks an appropriate mouse model for study. Here we developed an OLP-like mouse model using topical oxazolone to induce a delayed-type hypersensitivity-mediated oral lichenoid reaction. Histological and ultrastructural analysis confirmed hallmark pathological features of OLP, including band-like CD8+ T cell infiltration and basal cell damage, and the presence of Civatte bodies. Comparative transcriptomic analysis revealed significant similarity between RNA-seq profiles of the mouse model and human OLP lesions, highlighting shared upregulated genes and enriched pathways, particularly those related to IFN-γ signaling and cytotoxic T cell activity. Functional studies demonstrated that the OLP phenotype depended on IFN-γ, with local priming by IFN-γ intensifying the disease through upregulation of major histocompatibility complex class I. Additionally, the absence of Langerhans cells exacerbated disease severity in vivo. Therapeutic evaluation showed that the JAK inhibitors baricitinib and ruxolitinib effectively reduced disease burden and provided mechanistic insights. In conclusion, this OLP-like mouse model recapitulates key immunopathological and transcriptomic features of human OLP, offering a robust platform for dissecting disease mechanisms and evaluating novel therapeutic strategies.
Authors
Zhenlai Zhu, Tinglan Yang, Peng Peng, Kang Li, Wen Qin, Chen Zhang, Shuyan Wang, Yuanyuan Wang, Minghui Wei, Erle Dang, Meng Fu, Hao Guo, Wen Yin, Shuai Shao, Qing Liu
Abstract
Epigenetic macromolecular enzyme complexes tightly regulate gene expression at the chromatin level and have recently been found to colocalize with RNA splicing machinery during active transcription; however, the precise functional consequences of these interactions are uncertain. Here, we identify unique interactions of the CoREST repressor complex (LSD1-HDAC1-CoREST) with components of the RNA splicing machinery and their functional consequences in tumorigenesis. Using mass spectrometry, in vivo binding assays, and cryo-EM we find that CoREST complex-splicing factor interactions are direct and perturbed by the CoREST complex selective inhibitor, corin, leading to extensive changes in RNA splicing in melanoma and other malignancies. Moreover, these corin-induced splicing changes are shown to promote global effects on oncogenic and survival-associated splice variants leading to a tumor-suppressive phenotype. Using machine learning models, MHC IP-MS, and ELISpot assays we identify thousands of neopeptides derived from unannotated splice sites which generate corin-induced splice-neoantigens that are demonstrated to be immunogenic in vitro. Corin is further shown to reactivate the response to immune checkpoint blockade, effectively sensitizing tumors to anti-PD1 immunotherapy. These data position CoREST complex inhibition as a unique therapeutic opportunity which perturbs oncogenic splicing programs while also creating tumor-associated neoantigens that enhance the immunogenicity of current therapeutics.
Authors
Robert J. Fisher, Kihyun Park, Kwangwoon Lee, Katarina Pinjusic, Allison Vanasse, Christina S. Ennis, Parisa Farokh, Scott B. Ficarro, Jarrod A. Marto, Hanjie Jiang, Eunju Nam, Stephanie Stransky, Joseph Duke-Cohan, Melis A. Akinci, Anupa Geethadevi, Eric Raabe, Ana Fiszbein, Shadmehr Demehri, Simone Sidoli, Chad W. Hicks, Derin B. Keskin, Catherine J. Wu, Philip A. Cole, Rhoda M. Alani
Abstract
Fibroblast to myofibroblast transition is a critical event required for effective tissue repair. In pathologic wound repair processes, such as type 2 diabetes (T2D), fibroblast to myofibroblast transition is impaired. The exact factors that control this transition in wounds are unclear. Here, using human tissue and murine transgenic models, we show that the histone methyltransferase SETDB2 is elevated in diabetic wound fibroblasts and TNF-α represses fibroblast to myofibroblast transition via Setdb2. We identified that TNF-α increases Setdb2 in fibroblasts via a JAK1,3/STAT3 signaling pathway, where pharmacologic or genetic manipulation of this pathway altered Setdb2 in fibroblasts. We also found that fibroblasts treated with pro-inflammatory macrophage supernatants displayed increased Setdb2 and downregulated myofibroblast genes; inhibition of the TNF-α receptor reduced the upregulation of Setdb2. In diabetes, we showed that TNF-α signaling was increased in wound fibroblasts, which functions to increase Setdb2 expression and represses fibroblast to myofibroblast transition. Fibroblast-specific knockdown of SETDB2 and therapeutic inhibition of JAK1,3/STAT3 improved diabetic wound repair, where wound fibroblasts expressed increased myofibroblast genes. This study is the first to our knowledge to identify an epigenetic mechanism for reduced fibroblast to myofibroblast transition in diabetic wounds. Therapeutic targeting of the TNF-α/STAT3/SETDB2 axis in wound fibroblasts may improve diabetic wound healing.
Authors
Tyler M. Bauer, Kevin D. Mangum, Samuel D. Buckley, James Shadiow, Amrita D. Joshi, Christopher O. Audu, Jadie Y. Moon, Lindsey D. Hughes, Rachel Bogel, Lam C. Tsoi, Qinmennge Li, He Zhang, Steven Kunkel, Johann E. Gudjonsson, Frank M. Davis, Katherine A. Gallagher
No posts were found with this tag.