Muscle biology
Abstract
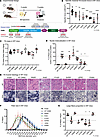
Centronuclear myopathies (CNMs) are rare congenital disorders characterized by muscle weakness, fiber hypotrophy, and organelle mislocalization. Most cases arise from mutations in MTM1 or DNM2, encoding myotubularin and dynamin-2, respectively. DNM2 is a GTPase that binds lipids, oligomerizes around membranes, and mediates fission. We previously showed that DNM2 levels are elevated in MTM1-CNM patients and Mtm1–/y mice, and that normalizing DNM2 rescues disease phenotypes. However, the specific DNM2 functions driving pathology remain unclear. Here, we expressed AAV-delivered WT and DNM2 mutants in WT and Mtm1–/y mouse muscles to disrupt specific DNM2 molecular functions. In WT mice, overexpression of WT DNM2 and most mutants induced CNM-like phenotypes, including reduced force, fiber hypotrophy, and centralized nuclei, consistent with gain-of-function mechanisms. The lipid-binding-defective mutant K562E did not induce disease-like phenotype. In Mtm1–/y mice, K562E mutant markedly improved muscle force, mass, and fiber size, while others failed to rescue. Therefore, we generated Mtm1–/y Dnm2K562E/+ mice, which showed full rescue of survival, motor function, and muscle force, with improved muscle mass, fiber size, and organelle positioning despite persistently elevated DNM2 levels. This study reveals that DNM2 lipid binding, not protein abundance or GTPase activity, drives pathology, and represents the most rational therapeutic target for DNM2 therapy in MTM1-CNM.
Authors
Raquel Gómez-Oca, Xènia Massana-Muñoz, David Reiss, Juliana De Carvalho Neves, Nadege Diedhiou, Roberto Silva-Rojas, Belinda S. Cowling, Marie Goret, Jocelyn Laporte
Abstract
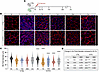
Skeletal muscle pathology is a critical but poorly understood contributor to neuromuscular degeneration in spinal and bulbar muscular atrophy (SBMA), a CAG/polyglutamine (polyQ) expansion disorder caused by mutation in the androgen receptor (AR). Using a gene-targeted SBMA mouse model, we applied single-nucleus RNA sequencing to identify a disease-specific population of skeletal muscle myonuclei that replaced normal myonuclear subtypes. This transition was associated with dysregulation of the pathway governed by PGC-1α, a central regulator of myofiber specification and metabolic identity. PGC-1α dysfunction in SBMA muscle was age-, hormone-, and polyQ length–dependent and was partially rescued by subcutaneous delivery of AR-targeted antisense oligonucleotides. Integrated ChIP-seq and RNA-seq analyses revealed that aberrant PGC-1α activity promoted the expression of a distinct set of myofiber specification genes while downregulating those that define healthy Type IIb and Type IIx myonuclei. We propose a model in which this dysfunction arose downstream of polyQ-mediated sequestration of PGC-1α cofactors MEF2, CREB, and CBP, leading to transcriptional reprogramming and cellular dysfunction. These findings implicated PGC-1α dysregulation as a key event linking AR polyQ expansion to skeletal muscle degeneration and suggested a shared mechanism for polyQ-mediated muscle pathology across related neurodegenerative diseases.
Authors
Curtis J. Kuo, Laura B. Chopp, Zhigang Yu, Luhan Ni, Hien T. Zhao, Janghoo Lim, Andrew P. Lieberman
Abstract
Lysophosphatidic acid (LPA) is a bioactive lipid that signals through G protein–coupled receptors (LPA1–6) and regulates multiple cellular processes, including fibrosis. Although LPA signaling has been implicated in fibrotic diseases in several organs, its role in skeletal muscle remains unclear. Here, we show that LPA/LPA1 signaling promotes fibrogenesis after sciatic nerve transection. Denervation induces differential expression of LPA signaling axis components and a transient early increase in intramuscular LPA levels. Pharmacological inhibition of LPA1/3 with Ki16425, or genetic deletion of LPA1, reduces extracellular matrix accumulation and expansion of fibro/adipogenic progenitors (FAPs) in denervated muscle. Although LPA blockade suppresses atrophy-related gene expression, it does not fully preserve myofiber size. Mechanistically, denervation increases YAP/TAZ expression, nuclear localization in FAPs, and transcriptional activity, effects that are attenuated by LPA axis inhibition. Furthermore, pharmacological inhibition of YAP/TAZ with verteporfin reduces fibrosis after denervation, supporting their role as critical downstream mediators. Finally, transient denervation activates the LPA axis, promotes muscle fibrosis, reduces axonal density in the sciatic nerve, and increases neuromuscular junction instability, effects reversed by Ki16425. Together, these findings identify the LPA/LPA1/YAP/TAZ pathway as a key driver of denervation-induced muscle fibrosis and a potential therapeutic target in neuromuscular disorders.
Authors
Meilyn Cruz-Soca, Adriana Córdova-Casanova, Jennifer Faundez-Contreras, Nicolás W. Martínez, Francesca Vaccaro-Rivera, Sebastián Bazaes-Astorga, Cristian Gutiérrez-Rojas, Felipe S. Gallardo, Daniela L. Rebolledo, Felipe A. Court, Jerold Chun, Carlos P. Vio, Soledad Matus, Juan Carlos Casar, Enrique Brandan
Abstract
X-linked myotubular myopathy (XLMTM) is a rare genetic disorder that typically presents at birth with progressive muscle weakness and respiratory difficulties and is caused by myotubularin-1 (MTM1) gene mutations. Here we examine the role of phosphatidylinositol-4-phosphate 3-kinase catalytic subunit type 2 beta (PIK3C2B), a lipid kinase that interacts with MTM1, in XLMTM in various models. We examined the effect of BLU3797, a novel, highly potent, selective, orally bioavailable PIK3C2B inhibitor, on survival, muscle development, myofiber phenotypes, and gene expression in MTM1-/y mice. PIK3C2B-deficient XLMTM animals demonstrated increased survival, restored muscle function, fewer myofibers with centralized nuclei, and normalization of disease-associated molecular markers. BLU3797 alleviated the XLMTM phenotype in a dose-dependent and reversible manner. Loss of functional PIK3C2B in XLMTM mice promoted a more differentiated, adult-like myofiber profile, which was strongly associated with normalization of disease surrogates and a reduction in markers of early muscle development and regeneration. BLU3797 treatment appears to modulate the expression of microRNAs associated with satellite cell activation and myofiber fusion. These findings indicate that PIK3C2B inhibition with BLU3797 effectively reverses the XLMTM disease phenotype by enhancing muscle function and promoting development toward a more mature state.
Authors
Andrew Shearer, Melissa L. Brooks, Maxine M. Chen, Thiwanka Samarakoon, John Hsieh, Gramoz Kondakci, Emanuele Perola, Jason Brubaker, Kristina Fetalvero, Stefanie Schalm, Joana Caetano-Lopes
Abstract
Limb-girdle muscular dystrophy R2 (LGMD R2) is an autosomal recessive disorder caused by dysferlin deficiency, leading to progressive muscle weakness and wasting. The lack of reliable clinical biomarkers has limited disease monitoring and therapeutic evaluation. Here, we identified Disabled-2 (DAB2) as a molecular and clinical indicator of disease state in LGMD R2. Transcriptomic profiling revealed a significant upregulation of DAB2 in induced pluripotent stem cell–derived (iPSC-derived) myotubes from patients, a finding validated in muscle biopsies from 14 dysferlin-deficient individuals and in dysferlin-deficient Bla/J mice, where DAB2 levels increased with disease progression. Importantly, AAV-mediated expression of full-length dysferlin restored DAB2 levels, supporting its value as a dynamic readout of disease activity for both disease monitoring and therapeutic response. Given the established role of DAB2 in clathrin-mediated endocytosis, particularly in LDL receptor internalization and cholesterol homeostasis, and the pathological lipid accumulation reported in LGMD R2, we investigated its contribution to lipid dysregulation. High DAB2 expression paralleled lipid deposition in patient muscles, iPSC-derived myotubes, and mouse tissue, whereas siRNA-mediated DAB2 knockdown reduced lipid accumulation in LGMD R2 myotubes. Collectively, these findings suggest that DAB2 functions as a mechanistic link between dysferlin deficiency, altered lipid handling, and disease severity, and they highlight its potential as a prognostic marker and therapeutic response measure for LGMD R2.
Authors
Celine Bruge, Nathalie Bourg, Emilie Pellier, Quentin Miagoux, Manon Benabides, Noella Grossi, Hassan Hayat, Margot Jarrige, Helene Polveche, Valeria Agostini, Anthony Brureau, Stephane Vassilopoulos, Teresinha Evangelista, Gorka Fernández-Eulate, Tanya Stojkovic, Isabelle Richard, Xavier Nissan
Abstract
Thyroid hormone signaling is an essential regulator of skeletal muscle development, function, and metabolism, yet the specific signaling pathways required for muscle regeneration are not yet defined. We used scRNA-seq and the FUCCI (fluorescent ubiquitination-based cell cycle indicator) reporter mouse model to examine how hypothyroidism impacts repair processes after cardiotoxin-induced injury in mice. During regeneration, and up to 2 months after injury, hypothyroid muscles displayed smaller myofibers and a shift to slower oxidative fiber types. scRNA-seq of tibialis anterior muscle during regeneration revealed that hypothyroidism reduced myogenic-lineage diversity. Cell cycle analysis confirmed delayed cell cycle progression at 5 and 14 days after injury, with skeletal muscle stem cells stalled at the G1/S transition, hindering differentiation. Transcriptomic data revealed altered nonmyogenic dynamics, including elevated activated fibro-adipogenic progenitors (FAPs) early in repair and persistent proinflammatory macrophages. Integrative regulon and ligand-receptor analysis further demonstrated that triiodothyronine acted through dual modes: a direct transcriptional control of myogenic cell cycle and oxidative programs and an indirect paracrine remodeling mediated by FAP and immune signaling networks. This study identified what we believe to be novel effects of hypothyroidism on myogenic heterogeneity and impaired tissue repair, offering insights into muscle-wasting mechanisms relevant to hypothyroidism-associated myopathy and sarcopenia.
Authors
Paola Aguiari, Valentina Villani, Yan-Yun Liu, Gianni Carraro, Gregory A. Brent, Laura Perin, Anna Milanesi
Abstract
Cachexia is a debilitating syndrome characterized by progressive skeletal muscle wasting, commonly affecting cancer patients, particularly those with pancreatic cancer. Despite its clinical significance, the molecular mechanisms underlying cancer cachexia remain poorly understood. In this study, we utilized single-nucleus RNA sequencing (snRNA-seq) and bulk RNA-seq, complemented by biochemical and histological analyses, to investigate molecular alterations in the skeletal muscle of the KPC mouse model of pancreatic cancer cachexia. Our findings demonstrated that KPC tumor growth induced myofiber-specific changes in the expression of genes involved in proteolytic pathways, mitochondrial biogenesis, and angiogenesis. Notably, tumor progression enhanced the activity of specific transcription factors that regulate the mTORC1 signaling pathway, along with genes involved in translational initiation and ribosome biogenesis. Skeletal muscle-specific, inducible inhibition of mTORC1 activity further exacerbated muscle loss in tumor-bearing mice, highlighting its protective role in maintaining muscle mass. Additionally, we uncovered new intercellular signaling networks within the skeletal muscle microenvironment during pancreatic cancer-induced cachexia. Together, our study revealed previously unrecognized molecular mechanisms that regulates skeletal muscle homeostasis and identified potential therapeutic targets for the treatment of pancreatic cancer–associated cachexia.
Authors
Bowen Xu, Aniket S. Joshi, Meiricris Tomaz da Silva, Silin Liu, Ashok Kumar
Abstract
This investigation leverages single-cell RNA sequencing (scRNA-Seq) to delineate the contributions of muscle-resident Schwann cells to neuromuscular junction (NMJ) remodeling by comparing a model of stable innervation with models of reinnervation following partial or complete denervation. The study discovered multiple distinct Schwann cell subtypes, including a novel terminal Schwann cell (tSC) subtype integral to the denervation-reinnervation cycle, identified by a transcriptomic signature indicative of cell migration and polarization. The data also characterizes three myelin Schwann cell subtypes, which are distinguished based on enrichment of genes associated with myelin production, mesenchymal differentiation or collagen synthesis. Importantly, SPP1 signaling emerges as a pivotal regulator of NMJ dynamics, promoting Schwann cell proliferation and muscle reinnervation across nerve injury models. These findings advance our understanding of NMJ maintenance and regeneration and underscore the therapeutic potential of targeting specific molecular pathways to treat neuromuscular and neurodegenerative disorders.
Authors
Steve D. Guzman, Ahmad Abu-Mahfouz, Carol S. Davis, Lloyd P. Ruiz, Peter C.D. Macpherson, Susan V. Brooks
Abstract
Duchenne muscular dystrophy (DMD) is a fatal genetic muscle-wasting disease characterized by loss of dystrophin protein. Therapeutic attempts to restore a functional copy of dystrophin to striated muscle are under active development, and many utilize adeno-associated viral (AAV) vectors. However, the limited cargo capacity of AAVs precludes delivery of full-length dystrophin, a 427 kDa protein, to target tissues. Recently, we developed a novel method to express large dystrophin constructs using the protein trans-splicing (PTS) mechanism mediated by split inteins and myotropic AAV vectors. The efficacy of this approach to restore muscle function in mdx4cv mice was previously assessed using histology, dystrophin immunolabeling, and western blotting. Here, we expand our molecular characterization of dystrophin constructs with variable lengths using a mass spectrometry-based proteomics approach, providing insight into unique protein expression profiles in skeletal muscles of wild-type, dystrophic mdx4cv, and AAV-treated mdx4cv. Our data reveal several affected cellular processes in mdx4cv skeletal muscles with changes in the expression profiles of key proteins to muscle homeostasis, whereas successful expression of dystrophin constructs results in an intermediate to complete restoration. This study highlights several biomarkers that could be used in future preclinical or clinical studies to evaluate the effectiveness of therapeutic strategies.
Authors
Erynn E. Johnson, Theodore R. Reyes, Jeffrey S. Chamberlain, James M. Ervasti, Hichem Tasfaout
Abstract
X-linked myotubular myopathy (XLMTM) due to MTM1 mutations is a rare and often lethal congenital myopathy. Its downstream molecular and cellular mechanisms are currently incompletely understood. The most abundant protein in muscle, myosin, has been implicated in the pathophysiology of other congenital myopathies. Hence, in the present study, we aimed to define whether myosin is also dysfunctional in XLMTM and whether it thus may constitute a potential drug target. To this end, we used skeletal muscle tissue from human patients and canine/mouse models; we performed Mant-ATP chase experiments coupled with X-ray diffraction analyses and LC/MS-based proteomics studies. In XLMTM humans, we found that myosin molecules are structurally disordered and preferably adopt their ATP-consuming biochemical state. This phosphorylation-related (mal)adaptation was mirrored by a striking remodelling of the myofibre energetic proteome in XLMTM dogs. In line with these, we confirmed an accrued myosin ATP consumption in mice lacking MTM1. Hence, we treated these, with a myosin ATPase inhibitor, mavacamten. After a four-week treatment period, we observed a partial restoration of the myofibre proteome, especially proteins involved in cytoskeletal, sarcomeric and energetic pathways. Altogether, our study highlights myosin inhibition as a new potential drug mechanism for the complex XLMTM muscle phenotype.
Authors
Elise Gerlach Melhedegaard, Fanny Rostedt, Charlotte Gineste, Robert A.E. Seaborne, Hannah F. Dugdale, Vladimir Belhac, Edmar Zanoteli, Michael W. Lawlor, David L. Mack, Carina Wallgren-Pettersson, Anthony L. Hessel, Heinz Jungbluth, Jocelyn Laporte, Yoshihiko Saito, Ichizo Nishino, Julien Ochala, Jenni Laitila
No posts were found with this tag.