Abstract
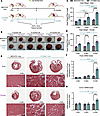
Acute kidney injury (AKI) is a common and fatal complication of severe pneumonia, yet the mechanisms linking pulmonary inflammation to remote kidney injury remain poorly understood. Multicenter cohort data (n = 300) revealed that the incidence of severe pneumonia–associated AKI (SP-AKI) was 53.6%, with a mortality rate of 24.2%. SP-AKI was associated with elevated circulating levels of HMGB1, NETs, and IL-33. Murine experiments demonstrated that alveolar HMGB1 triggers the formation of IL-33–enriched NETs, which migrate to the kidney and activate tubular ST2/NF-κB signaling, driving inflammation and apoptosis. Genetic knockout of IL-33, ST2, or the NET-forming key enzyme PAD4, as well as pharmacological inhibition of HMGB1, IL-33, or NETs, all attenuated lung and kidney injury. Exogenous HMGB1 amplified NET-mediated IL-33 release, establishing a self-sustaining HMGB1/NET/IL-33 feed-forward loop. PAD4 deficiency completely blocked NET generation and disrupted HMGB1/IL-33 signaling. This study identified and validated a damage-associated molecular pattern–driven (DAMP-driven) HMGB1/NET/IL-33 signaling axis that mediates remote kidney injury in SP-AKI, redefining NETs from local effectors to cross-organ pathogenic carriers, thereby providing potential DAMP-targeted therapeutic avenues for SP-AKI.
Authors
Mengqing Ma, Hao Zhang, Weijuan Deng, Xia Du, Mengxing Chen, Dawei Chen, Binbin Pan, Zhaowei Wang, Ting Chen, Caimei Chen, Xin Wan, Changchun Cao
Abstract
A large interindividual variability in weight loss outcomes following bariatric surgery is reported. To ensure optimal management of patients, it is crucial to accurately identify candidates most likely to benefit the most from the intervention. Since genetic variants largely contribute to surgery response, polygenic scores (PGS) derived from genome-wide association studies (GWAS) could constitute valuable tools for clinical decision making. We developed and evaluated PGS to predict the weight loss response in 540 patients with a body mass index (BMI) of 35 kg/m2 or higher who underwent biliopancreatic diversion with duodenal switch. Summary statistics derived from BMI-derived GWAS, together with summary statistics from previously published GWAS of BMI and adiposity features, were used to construct, evaluate, and benchmark weight loss PGS. The full-adjusted BMI PGS model built in the entire cohort explained 39.6% of the mean-over-time excessive body weight loss (%EBWL), while the BMI-PGS built in the training dataset explained 38.9%. All benchmarked PGS based on BMI showed a significant relationship with mean-over-time %EBWL. These findings highlight the potential of BMI PGS in predicting weight loss after bariatric surgery and support their use as promising tools to improve the effectiveness of future antiobesity treatments.
Authors
Bastien Vallée Marcotte, Juan de Toro-Martín, André Tchernof, Louis Pérusse, Simon Marceau, Marie-Claude Vohl
Abstract
Thoracic aortic aneurysm and dissection (TAAD) involves a progressive dilation of the aortic wall associated with degradation of the extracellular matrix (ECM), cystic medial degeneration, smooth muscle cell (SMC) dysfunction, and rarefaction. TAAD etiology and pathogenesis suggest that alteration of mechanical force propagation may contribute to SMC dysfunction. This study aims to determine the role of SMC focal adhesion proteins, which are key components of force transmission, in TAAD pathogenesis. scRNA-seq analysis of human TAA aortas showed reduced expression of intracellular focal adhesion components, including PTK2 (FAK), VCL, ILK, and TES transcripts, in SMCs. Additionally, protein levels of FAK, ILK, and VCL were decreased in the aorta of patients with TAA. SMC-specific Ptk2, Vcl, and Ilk KO mice treated with β-aminopropionitrile (BAPN) exhibited increased mortality, aortic dilation, ECM breakdown, and SMC loss. Mechanistically, knocking down FAK, ILK, and VCL exacerbated gliotoxin-induced SMC anoikis, whereas overexpressing full-length WT and dead-kinase FAK conferred resistance to apoptosis and cell detachment, indicating that FAK’s protective effects depend on its expression rather than its enzymatic activity. Inhibition of FAK kinase activity did not affect SMC apoptosis in vitro or aortic dilation in vivo. Our findings demonstrate that the expression of focal adhesion proteins protects against TAAD progression and SMC anoikis independently of FAK kinase activity.
Authors
Zhenyuan Zhu, Mingjun Liu, Jianxin Wei, Deepa Suryanarayan, Parya Behzadi, Robert Edgar, Julie A. Phillippi, Cynthia St. Hilaire, Cristina Espinosa-Diez, Delphine Gomez
Abstract
Sleep disturbance is a prevalent yet poorly understood comorbidity in autism spectrum disorders (ASD). Here, we uncover a bidirectional regulatory axis connecting the ASD risk gene POGZ to core circadian mechanisms. We demonstrate that Pogz is widely expressed in the suprachiasmatic nucleus (SCN), the central pacemaker of the circadian rhythms, and exhibits circadian oscillations in both the hypothalamus and liver, with its transcription directly regulated by the circadian molecule DBP through a D-box element in its proximal enhancer. Pogz-deficient mice exhibited prolonged circadian periodicity, impaired light-induced phase shift, delayed adaption to an 8-hour advance jet-lag, and reduced SCN c-Fos activation in response to light pulses. Mechanistically, POGZ interacts with and enhances the transcription activity of CREB, a key regulator of light-induced phase resetting. Notably, Pogz deletion leads to ASD-related deficits in social novelty and cognition, with cognitive impairments influenced by both photoperiod and behavioral paradigm. Our findings, thus, reveal a critical, previously unrecognized intersection between an ASD risk gene and circadian clock, offering insights into the pathogenesis of core ASD symptoms and comorbid sleep disturbances.
Authors
Ting Wu, Jiao He, Chu-Jun Xu, Chi-Yu Li, Pingchuan Zhang, Yanfeng Wang, Shanshan Zhu, Lusi Zhang, Jingtan Zhu, Jing Zhang, Jia-Da Li, Huadie Liu
Abstract
We previously reported that excessive angiotensin-II→AT receptor-1 (AT→ATR1) signaling results in sickle cell anemia–associated (SCA-associated) nephropathy. Herein, we showed that hyperangiotensinemia in SCA results from high erythroid cell–generated reactive oxygen species (ROS), which oxidized angiotensinogen (ATGN) and favored its rapid conversion to AT. Increased AT→ATR1 signaling in SCA erythroid cells generated ROS and created a positive feedback loop of ROS→oxidized ATGN→AT→ATR1→ROS, perpetuating the hyperangiotensinemia. ATR1 blocker, losartan, reduced erythrocyte ROS, oxidized ATGN, and AT levels. The ROS→AT→ATR1→ROS loop was driven by sickle erythropoiesis, as it was reproduced when WT mice were transplanted with SCA hematopoiesis. Using SCA and WT mice with germline- and erythroid-specific ATR1 deficiency, we found that stress erythropoiesis, but not steady-state erythropoiesis, was critically dependent on erythroid AT→ATR1 signaling, which acted in harmony with increased erythropoietin signaling. Furthermore, instead of the canonical AT→ATR1→NADPH-oxidase→ROS signaling in steady-state erythropoiesis, AT→ATR1 signaling in stress erythroid cells increased mitochondrial mass and dysfunctional mitochondria, which thereby increased ROS. SCA mice with erythroid-specific ATR1 deficiency had decreased RBC accumulation of dysfunctional mitochondria and decreased ROS, which reduced SCA-associated nephropathy. Overall, we demonstrate that AT→ATR1 signaling was essential for stress erythropoiesis but led to increased dysfunctional mitochondria retention in mature RBCs, which generated ROS and perpetuated hyperangiotensinemia, resulting in end-organ damage.
Authors
Parul Rai, Swarnava Roy, Paritha Arumugam, Diamantis G. Konstantinidis, Sithara Raju Ponny, Marthe-Sandrine Eiymo Mwa Mpollo, Archana Shrestha, Theodosia A. Kalfa, Punam Malik
Abstract
Glioblastoma (GBM) is the most malignant primary brain tumor. The presence of glioma stem/initiating cells (GICs) is known to cause strong treatment resistance; therefore, GICs are a major target for GBM therapy, although there are no therapies targeting GICs clinically. To identify novel treatments for GBMs, we performed drug repurposing screening using GICs and identified the T-type calcium channel blocker lomerizine — a migraine prophylactic drug. Lomerizine inhibited proliferation, migration, invasion, and cell cycle progression and induced apoptosis in GICs and differentiated glioma cells. Lomerizine had antitumor effects by inactivating STAT3 in all cell lines. Furthermore, lomerizine also dephosphorylated AKT and ERK only in GICs and had strong tumor-suppressive ability. Lomerizine also reduced tumor volume and prolonged overall survival in vivo. Based on our data from in vitro and in vivo experiments, lomerizine has potential as a GBM therapeutic agent targeting both GICs and differentiated glioma cells and could benefit GBM patients.
Authors
Toshiya Ichinose, Sho Tamai, Nozomi Hirai, Takashi Maejima, Kosuke Nambu, Hemragul Sabit, Shingo Tanaka, Masashi Kinoshita, Masahiko Kobayashi, Michihiro Mieda, Atsushi Hirao, Mitsutoshi Nakada
Abstract
Allogeneic islet transplantation is an effective treatment for type 1 diabetes, but its clinical use is limited by rejection involving innate and adaptive immune responses, requiring lifelong immunosuppression. We herein engineered islets that transiently display 2 immunomodulators chimeric with streptavidin (SA), thrombomodulin (SA-TM) and CD47 (SA-CD47), for localized modulation of both innate and adaptive immune responses. The engineering process did not impact islet viability, glucose responsiveness, and metabolic activity. Intraportal transplantation into allogeneic recipients achieved sustained survival, with 8 out of 11 grafts surviving 120–330 days without immunosuppression. In contrast, non-engineered islets were acutely rejected (median survival time [MST] = 12 days), while islets engineered with SA-TM showed delayed rejection (MST = 13.5 days) and those with SA-CD47 exhibited prolonged survival (MST = 24 days). Double-engineered islets generated a localized tolerogenic immune environment characterized by low frequencies of inflammatory innate immune cells and increased frequencies of M2 macrophages, myeloid-derived suppressor cells, and CD4+FoxP3+ T regulatory cells. The transcriptomic analysis showed downregulation of proinflammatory and upregulation of immune regulatory pathways. Our results demonstrate that transient co-display of immunomodulatory molecules on the islet surface is a versatile platform with significant translational potential for islet transplantation.
Authors
Shadab Kazmi, Mohammad Tarique, Darshan Badal, Vahap Ulker, Ali Turan, Kathleen M. Yee-Flores, Abdalmonam Jadou Nejma, Esma S. Yolcu, Haval Shirwan
Abstract
Myotonic dystrophy type 1 (DM1) is caused by an expanded CTG repeat in the DMPK gene, resulting in mutant transcripts that form expanded CUG (CUGexp) RNA foci and sequester muscleblind-like (MBNL) RNA-binding proteins. DM1 is multisystemic, with progressive worsening of disease manifestations in affected tissues. Disease progression is attributed to somatic expansion of the CTG repeats with age, resulting in production of CUGexp RNA with enhanced intrinsic toxicity due to increased MBNL sequestration. To determine the degree to which cardiac disease progression can occur independently of repeat expansion, we used a transgenic DM1 mouse model with inducible heart-specific expression of a stable, interrupted 960-CUG-repeat RNA. Sustained CUGexp RNA expression caused progressive cardiac enlargement, contractile dysfunction, conduction delay, myocardial fibrosis, and reduced survival, while MBNL-dependent splicing defects remained static, consistent with the stable repeat length. We also determined the degree of reversibility after different periods of CUGexp RNA expression by shutting off the repeat-containing transgene. Suppression of CUGexp RNA expression rescued cardiac abnormalities, but reversibility declined with longer exposure to the toxic RNA. These findings demonstrate that prolonged expression of stable CUGexp RNA drives progressive cardiac pathology, revealing a mechanism of disease progression in DM1 in addition to somatic expansion.
Authors
Rong-Chi Hu, Mohammadreza Tabary, Xander H.T. Wehrens, Thomas A. Cooper
Proteomic analyses of human islets reveal potential markers of β cell dysfunction during prediabetes
Abstract
The mechanisms driving progressive β cell dysfunction in type 2 diabetes remain incompletely understood. This study aimed to identify pancreatic islet proteome changes that could predict diabetes onset. We isolated islets from individuals without diabetes undergoing partial pancreatectomy, previously characterized for glucose tolerance, insulin sensitivity, and insulin secretion, using laser capture microdissection, and analyzed them via high-performance liquid chromatography–mass spectrometry. Proteomic analysis revealed that individuals with impaired glucose tolerance (IGT) had reductions in proteins regulating glycolysis (PGK1, G3P), lipid metabolism (ACBP, ARF1), glucose transport (14-3-3B), and insulin secretion (STARD10, CAPDS) compared with normal glucose-tolerant (NGT) individuals. Additionally, IGT islets showed impaired expression of proteins involved in glucose- and incretin-stimulated insulin response (CREB1, IQGA1). Stratification by β cell glucose sensitivity (βGS) indicated that individuals with lower βGS exhibited reduced levels of insulin maturation (ERO1B) and antiapoptotic proteins (CASP8, PAK2, SKP1), along with increased SEL1L, a factor promoting endocrine precursor differentiation. These findings suggest that early defects in glucose metabolism and insulin secretion characterize IGT, while reduced βGS may trigger compensatory mechanisms, through enhanced β cell survival or neogenesis, to delay type 2 diabetes progression. Overall, proteomic alterations in prediabetic islets provide potential early predictive markers and targets for interventions aimed at preserving β cell function.
Authors
Chiara Maria Assunta Cefalo, Teresa Mezza, Giuseppe Quero, Sergio Alfieri, Donatella Lucchetti, Filomena Colella, Alessandro Sgambato, Wei-Jun Qian, Andrea Mari, Alfredo Pontecorvi, Andrea Giaccari, Rohit N. Kulkarni
Abstract
The molecular mechanisms responsible for the “atopic march” of allergic skin disease to allergic airway disease are incompletely understood. Secreted phospholipase A2 group X (sPLA2-X) is implicated in human asthma and modulates airway hyperresponsiveness (AHR) and inflammation in murine models of allergic asthma. We developed a complete proteolytic allergen model of dermal sensitization followed by airway challenge to mimic the “atopic march” and examined the role of sPLA2-X in regulating peripheral allergen sensitization, AHR, and airway inflammation. Pla2g10–/– mice receiving both house dust mite (HDM) peripheral sensitization and airway challenge had attenuated AHR relative to WT mice and lower airway eosinophils. Transgenic C57BL/6 hPLA2G10 mice (only expressing the human sPLA2-X gene) receiving treatment with a small molecule inhibitor of sPLA2-X (ROC0929) during the dermal sensitization phase demonstrated attenuated AHR and a reduction HDM-specific tissue-resident memory CD4+ T cells in the lung. Thus, sPLA2-X acts as an endogenous adjuvant to facilitate allergic sensitization in the periphery, which leads to AHR and airway inflammation following inhalation of the allergen. These results provide proof of concept that inhibition of sensitization in the periphery with a sPLA2-X inhibitor modulates subsequent allergen-induced airway dysfunction.
Authors
Ryan C. Murphy, Ying Lai, Yu-Hua Chow, Matt Liu, Brian D. Hondowicz, Dowon An, Marion Pepper, William A. Altemeier, Teal S. Hallstrand
No posts were found with this tag.