Review
Open Access |  10.1172/jci.insight.199953
10.1172/jci.insight.199953
A double-edged sword: iron regulation in alveolar lung epithelial repair
Ugonna Mbaekwe,1,2 Sarah Kenny,3 Suzanne M. Cloonan,3 and Corrine R. Kliment1,2
1Division of Pulmonary, Allergy, Critical Care Medicine, and
2Department of Cellular and Molecular Pathology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA.
3School of Medicine, Trinity Biomedical Sciences Institute, Trinity College Dublin, Dublin, Ireland.
Address correspondence to: Corrine Kliment, University of Pittsburgh School of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, 200 Lothrop Street, Biomedical Science Tower W1254, Pittsburgh, Pennsylvania 15213, USA. Email: ckliment@pitt.edu.
Find articles by Mbaekwe, U. in: PubMed | Google Scholar
1Division of Pulmonary, Allergy, Critical Care Medicine, and
2Department of Cellular and Molecular Pathology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA.
3School of Medicine, Trinity Biomedical Sciences Institute, Trinity College Dublin, Dublin, Ireland.
Address correspondence to: Corrine Kliment, University of Pittsburgh School of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, 200 Lothrop Street, Biomedical Science Tower W1254, Pittsburgh, Pennsylvania 15213, USA. Email: ckliment@pitt.edu.
Find articles by
Kenny, S.
in:
PubMed
|
Google Scholar
|

1Division of Pulmonary, Allergy, Critical Care Medicine, and
2Department of Cellular and Molecular Pathology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA.
3School of Medicine, Trinity Biomedical Sciences Institute, Trinity College Dublin, Dublin, Ireland.
Address correspondence to: Corrine Kliment, University of Pittsburgh School of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, 200 Lothrop Street, Biomedical Science Tower W1254, Pittsburgh, Pennsylvania 15213, USA. Email: ckliment@pitt.edu.
Find articles by
Cloonan, S.
in:
PubMed
|
Google Scholar
|

1Division of Pulmonary, Allergy, Critical Care Medicine, and
2Department of Cellular and Molecular Pathology, University of Pittsburgh School of Medicine, Pittsburgh, Pennsylvania, USA.
3School of Medicine, Trinity Biomedical Sciences Institute, Trinity College Dublin, Dublin, Ireland.
Address correspondence to: Corrine Kliment, University of Pittsburgh School of Medicine, Division of Pulmonary, Allergy, Critical Care and Sleep Medicine, 200 Lothrop Street, Biomedical Science Tower W1254, Pittsburgh, Pennsylvania 15213, USA. Email: ckliment@pitt.edu.
Find articles by
Kliment, C.
in:
PubMed
|
Google Scholar
|

Published May 8, 2026 - More info
JCI Insight. 2026;11(9):e199953. https://doi.org/10.1172/jci.insight.199953.
© 2026 Mbaekwe et al. This work is licensed under the Creative Commons Attribution 4.0 International License. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
-
Abstract
The oxygen-rich milieu of the lungs necessitates precise iron homeostasis and regulation, processes that are fundamental to pulmonary physiology but often receive limited attention. However, in recent years, dysregulation of iron homeostasis has been linked to numerous acute and chronic respiratory diseases. Here, we comprehensively evaluate the mechanisms governing iron homeostasis in the alveolar epithelium of the lung and examine how iron dysregulation contributes to impaired alveolar epithelial repair in respiratory disease. This Review focuses on the effects of iron on alveolar epithelial cell homeostasis and repair and disease pathogenesis. There will be a focus on emerging interventions designed to reestablish iron homeostasis and their potential therapeutic implications related to enhancing lung repair and limiting the progression of lung disease.
-
The alveolar epithelium
The lungs maintain the largest surface area of any organ in the human body, ranging from 70 to 100 square meters (1), and are constantly exposed to external damaging stimuli, which can cause injury and subsequent disease. The lung has inherent repair processes in place to respond to damage and promote regeneration. The alveolar epithelium is a specialized layer of epithelial cells crucial for gas exchange and lung health. Alveoli are composed of a monolayer of flattened, squamous alveolar type 1 (AT1) cells that cover 95% of the surface and cuboidal alveolar type 2 (AT2) cells (2). AT2 cells produce surfactant to reduce surface tension, preventing lung collapse during exhalation. AT2 cells also serve as self-renewing progenitors that, after lung injury, differentiate into AT1 cells, which regulate gas exchange and ion transport (3).
Other less abundant cell types, including mesenchymal, endothelial, and immune cells, contribute to the dynamic and spatially organized alveolar structure and defense (4–6) (Figure 1). Fibroblasts produce and remodel the ECM and alveolar architecture (6). Alveolar macrophages (AMs) serve as crucial immune cells that combat inhaled pathogens through phagocytosis and respond to environmental signals to stave off persistent inflammation and infection (7, 8). While these cell types are important in lung function, this Review will primarily focus on alveolar epithelial cells.
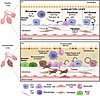 Figure 1
Figure 1AT2 dysfunction in the lung. A schematic overview of the main interactions and signaling pathways involved in AT2 self-renewal, proliferation, and differentiation into AT1 cells. In a self-renewing state, WNT canonical and FGFR/AKT pathways primarily maintain AT2 identity and function. During differentiation, AT2 cell transition is marked by Krt8+ cells (also called damage-associated transient progenitor [DATP] cells). In this state, metabolic changes occur, along with integrated stress responses (ISR) and increased Notch signaling. Activation of YAP/Taz signaling promotes differentiation, and the secretion of TGF-β and IL-1β from AT2 cells in specific contexts can interact with macrophages and fibroblasts to produce factors, such as FGF7 and FGF10, that support AT2 identity. Macrophages release cytokines, such as IL-1β and TNF-α, to enhance AT2 cell proliferation and differentiation. Disruptions in the alveolar epithelial repair process can cause pathological outcomes, including bronchiolization of the alveoli, immune infiltration, increased barrier permeability, failed repair and regeneration, and the development of senescence and fibrosis within the alveolar space. Created in BioRender.
-
Alveolar epithelium renewal and repair processes
Effective alveolar repair after lung injury is characterized by resolution of inflammation, reestablishment of the alveolar-capillary barrier, and restoration of gas exchange capability. Repair depends on timely AT2 cell proliferation for self-renewal and differentiation, which is regulated by intrinsic transcriptional factors and niche signals (2, 9–11) (Figure 1). FGFR2/AKT promotes AT2 cell survival and lineage maintenance. Loss of FGFR2 signaling in AT2 cells caused increased apoptosis, followed by compensation from remaining progenitor-like cells (12). Canonical WNT signaling maintains a WNT-responsive AT2 subpopulation (AXIN2+ alveolar epithelial progenitors) that preserves stemness during homeostasis and drives regenerative expansion after injury (13–15). Transcription factors, including thyroid transcription factor 1 (NKX2-1) and CEBPA, are critical for maintaining the identity, plasticity, and function of AT2 cells. NKX2-1 establishes and maintains AT2 cell fate by directly activating surfactant protein genes and repressing abnormal differentiation pathways. In addition, CEBPA is similarly enriched in mature AT2 cells, working in concert with NKX2-1 to maintain AT2 identity and to act as a gatekeeper, restricting activation of repair or transitional programs during stress (13, 16, 17).
AT2-to-AT1 differentiation is a multistep process involving both internal signaling, such as Notch signaling, and external mechanical cues that activate effectors like YAP/TAZ signaling. In AT2 cells, Notch signaling is instrumental in AT2 maintaining cell identity by promoting AT2-associated gene expression and preventing premature differentiation. At the onset of differentiation, Notch receptors undergo transcriptional suppression, thereby removing their inhibitory effect on AT1-related genes (18). During lung injury, increased tissue tension can induce dephosphorylation of YAP/TAZ, leading to nuclear translocation and subsequent activation of epithelial gene programs. These programs facilitate AT2 cell flattening and increase the expression of AT1 markers (19–21).
A complex interaction among the alveolar niche, epithelial progenitor cells, and various interstitial cells directs alveolar maintenance and repair. These supporting cells influence AT2 cell behavior through paracrine signaling and direct cell-cell interactions, modulating proliferation, differentiation, and survival during regeneration (22, 23). AT2 cells communicate with macrophages and fibroblasts through the secretion of context-dependent paracrine signals (Figure 1), such as TGF-β and IL-1β (4, 24–26). These cytokines stimulate fibroblasts to produce growth factors FGF7 and FGF10, thereby supporting AT2 cells (27–29). Additionally, secreted cytokines can induce macrophages to release IL-1β and TNF-α for further promotion of AT2 cell proliferation (30, 31). Consequently, continuous feedback loops are established, facilitating intricate multicellular interactions. Although these dynamics are important, the inherent properties of epithelial progenitor cells also significantly influence AT2 cell functioning and repair.
Alveolar repair requires substantial energy expenditure. Accordingly, AT2 cells exhibit a higher mitochondrial density and lower turnover than other lung cells, underscoring their reliance on mitochondrial function and energy metabolism (32, 33). There is growing evidence that disruptions in mitochondrial and metabolic pathways, such as calcium uptake, glutamine metabolism, glycolysis, and fatty acid oxidation, can lead to abnormal repair mechanisms and adversely affect AT2 cell reparative capacity, proliferation, and fate determination (34–38). Current evidence indicates that activation of the integrated stress response (ISR) may facilitate the transition of AT2 cells to AT1 cells (31, 38).
-
Dysregulation of alveolar renewal and repair
Disruption of alveolar epithelial repair mechanisms leads to pathological outcomes, such as chronic lung disease. Homeostatic lung repair is tightly regulated through a dynamic interplay of epithelial progenitor activation, mesenchymal remodeling, immune resolution, and vascular regeneration — each precisely orchestrated to restore tissue architecture after injury. These processes can be subverted by persistent injury, aging, or abnormal signaling, leading to maladaptive repair.
Key features of dysregulated alveolar repair include loss of AT2 cell identity, impaired AT2 proliferation, and blockade of AT2-to-AT1 differentiation. Other key features include the emergence of keratin 8+ (Krt8+) transitional cells (also called damage-associated transient progenitor [DATP] cells) derived from AT2 cells (Figure 1). Although AT2 cells are the primary regenerative stem cells, other epithelial cells, such as club cells, contribute to alveolar regeneration (39, 40). DATP cells are generally temporary and essential for effective alveolar epithelial regeneration. DATP cells are typically recognized by increased keratin expression, a partial loss of standard AT2 markers, and partial activation of AT1 cell programs (41). Several mechanistic regulators are increasingly recognized as driving and sustaining this transitional state, including TGF-β, IL-1β, HIF1α, ISR, and senescence pathways (31, 38, 42).
However, DATP cell persistence is linked to failed regeneration and fibrosis. In mouse bleomycin (BLM) lung injury models and humans with idiopathic pulmonary fibrosis (IPF), Krt8+ cells exhibit a senescence-like profile, high stress-response gene expression, and poor differentiation into AT1 cells (31, 42). AT2 cells can undergo cellular senescence, a cell cycle arrest process in response to stressors marked by increased levels of classical cell cycle arrest markers, Cdkn1a and Cdkn2a (24). Senescence further contributes to failed regeneration by promoting a senescence-associated secretory phenotype, characterized by secretion of proinflammatory factors, profibrotic mediators, and MMPs (43).
Ectopic expansion of airway-derived basal cells into the distal alveolar space indicates maladaptive alveolar repair. After severe injury — like H1N1 influenza or fibrosis — basal-like progenitors with Tp63 and Krt5 markers migrate into alveoli, forming abnormal structures that lack surfactant proteins and cannot support gas exchange (44). Airway secretory cells can dedifferentiate into Tp63+ basal-like progenitors, aiding alveolar regeneration during injury. However, dysregulation may cause bronchiolized, nonfunctional remodeling (45, 46). Basal cell–derived progenitors can act as emergency patches after injury, but their persistence causes maladaptive repair, displacing native alveolar progenitors and promoting pathological remodeling in chronic lung disease.
Disruption of these remodeling mechanisms, as occurs in chronic injury or aging, compromises repair fidelity, leading to abnormal lung regeneration with persistence of transitional cell states (31, 42), senescence accumulation (29, 47, 48), and failed epithelial restoration. Aberrant repair leads to pathological remodeling in chronic lung diseases like IPF and chronic obstructive pulmonary disease (COPD). Recent studies (49–54) identify iron homeostasis as a key regulator of alveolar repair, with iron imbalance driving oxidative stress, inflammation, senescence, and epithelial dysfunction that impair regeneration. This Review examines how iron influences epithelial renewal within the alveolar microenvironment.
-
Iron homeostasis and regulation
Iron is an essential metal that supports multiple processes, including oxygen transport (48), DNA repair, and synthesis of heme and iron-sulfur (Fe-S) clusters, with approximately 2% of human genes encoding an iron-protein (55). However, excess free labile iron is highly toxic owing to the production of ROS (56). Thus, iron’s function is a double-edged sword, emphasizing the importance of maintaining iron balance at both systemic and cellular levels.
Systemic iron regulation involves a multiorgan network overseeing absorption, recycling, and distribution to maintain balance (Figure 2). In mammals, iron is obtained from the diet and recycled internally, mainly absorbed in the duodenum as heme and nonheme iron (57). Heme iron is transported across the plasma membrane by heme carrier protein 1 (HCP1) (58) and broken down internally by heme-oxygenase 1 (HMOX1) (59, 60). Nonheme iron is reduced from ferric (Fe3+) to ferrous (Fe2+) iron by duodenal cytochrome B and imported via divalent metal transporter 1 (DMT1) (57). Iron is exclusively exported by ferroportin (FPN), while ferroxidases such as hephaestin and ceruloplasmin oxidize iron to facilitate binding to plasma transferrin and transport to other tissues (61). Macrophages recycle iron from senescent erythrocytes in the spleen, providing a significant source of iron (62). Hepatocytes regulate iron balance via hepcidin (HAMP)/FPN, adjusting distribution as needed. HAMP, a liver hormone, binds FPN, leading to its degradation and decreasing iron release and serum iron levels (63, 64).
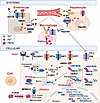 Figure 2
Figure 2General iron homeostasis. A schematic diagram showing systemic (top) and cellular iron regulation (bottom). Systemic iron control involves various organs managing absorption, recycling, and distribution to keep levels balanced. In mammals, iron is mainly absorbed in the duodenum as both heme and nonheme iron and then transported across cell membranes and transferred in the blood to other tissues. Macrophages and hepatocytes are vital for recycling iron from red blood cells and adjusting systemic iron levels. Cellular iron regulation includes controlling iron import, export, storage, and use. Low oxygen levels and iron deficiency activate transcription factors and regulatory proteins to boost iron absorption, while iron overload triggers protective pathways to sequester, detoxify, and maintain balance. The key proteins discussed in this Review are shown at the bottom of the figure, including heme carrier protein 1 (HCP1), heme-oxygenase 1 (HMOX1), duodenal cytochrome B (DCYTB), divalent metal transporter 1 (DMT1), ferroportin (FPN), ceruloplasmin (CP), hephaestin (HEPH), transferrin receptor 1 (TfR1), lipocalin (LCN2), poly(rC)-binding protein 1 (PCBP1), mitoferrin-1 and mitoferrin-2 (MFRN1/2), leukemia virus subgroup C cellular receptor 1 (FLVCR1), hypoxia-inducible factor 2 α (HIF2α), hypoxia response element (HRE), iron-sensing E3 ligase (FBXL5), iron regulatory proteins (IRP), nuclear receptor coactivator 4 (NCOA4), and nuclear factor erythroid 2–related factor 2 (NRF2). Created in BioRender.
Cellular iron homeostasis involves the regulation of iron import, export, storage, and utilization (Figure 2). Iron import involves several different mechanisms. Transferrin-bound iron is taken up through transferrin receptor 1 (TfR1) (65), which mediates receptor-dependent endocytosis followed by iron reduction by STEAP3 and endosomal iron release through DMT1 (66). Non-transferrin-bound iron can be imported through divalent metal transporters, such as DMT1 and ZIP family members ZIP8 and ZIP14, which import iron in metabolically active and inflammatory contexts (67, 68). HCP1 internalizes heme iron, while SLC22A17 mediates the uptake of lipocalin-bound (LCN2-bound) iron during immune activation (69). Iron export is mediated by FPN, while iron storage is primarily achieved through ferritin, which buffers iron. Efficient iron delivery to ferritin is accomplished by the cytosolic iron chaperone poly(rC)-binding proteins (70). Iron utilization is primarily driven by mitochondrial processes. Mitochondrial iron import is mediated by mitoferrin-1 and mitoferrin-2 (MFRN1/2) (49, 71, 72), which deliver iron for heme biosynthesis, Fe-S cluster assembly, and metabolic enzyme function. Excess heme can be exported from the cell via feline leukemia virus subgroup C cellular receptor 1 (FLVCR1) (73, 74) to maintain heme-iron balance.
Cellular iron levels are regulated at the level of gene expression. During hypoxia or iron deficiency, stabilization of HIF2α (75) activates transcription of hypoxia response element–containing genes that enhance iron uptake and utilization. Ubiquitin ligase HERC2 posttranscriptionally degrades iron-sensing ligase (FBXL5), leading to stabilization of iron regulatory proteins (IRPs) (76). In low iron states, IRP1 lacks its Fe-S cluster and switches to its iron response element–binding (IRE-binding) form (77). IRP1 stabilizes iron import protein-encoding mRNAs and represses translation of ferritin to reduce iron storage (78). IRP2, normally degraded by FBXL5, stabilizes when FBXL5 is degraded, increasing iron uptake and reducing storage (79, 80). Nuclear receptor coactivator 4–mediated (NCOA4-mediated) ferritinophagy is enhanced to liberate stored iron through ferritin degradation in lysosomes (81). Conversely, in conditions of iron overload, activation of the nuclear factor erythroid 2–related factor 2 antioxidant pathway induces the expression of genes involved in iron sequestration, detoxification, and redox defense., FBXL5 is posttranscriptionally stabilized and promotes degradation of IRPs. When bound to a Fe-S cluster, IRP1 converts into a cytosolic aconitase and loses the ability to bind IREs. IRP2 is degraded by FBXL5, while HERC2 degrades NCOA4 to limit ferritinophagy (82).
Tight regulation of cellular iron is important for meeting metabolic needs and controlling redox balance (Figure 3). Unregulated Fe2+ reacts with ROS through a Fenton reaction to generate highly reactive hydroxyl radicals (83–85) and catalyzes the oxidation of polyunsaturated fatty acids in membranes to form lipid hydroperoxides (86). This process sensitizes cells to an iron-dependent form of cell death called ferroptosis. Ferroptosis is driven by the lethal accumulation of lipid peroxides (87). When antioxidant defenses, especially glutathione peroxidase 4 (GPX4), are overwhelmed or impaired, lipid peroxides accumulate unchecked, triggering cell death. Ferroptosis is particularly relevant in tissues with high oxidative stress and iron turnover, like the lung, where dysregulated redox balance and iron overload contribute to both acute and chronic injury (88).
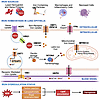 Figure 3
Figure 3Lung iron homeostasis. A schematic illustrating lung iron sources and regulation (top) and how iron dysregulation triggers ferroptosis (bottom) in lung epithelial cells affected by disease. The lung depends on endogenous sources, like cellular recycling, and exogenous sources, such as particulate matter, which increases oxidative stress and inflammation. Excess iron results in ROS, lipid peroxidation, and ferroptosis when antioxidants are overwhelmed. Created in BioRender.
-
Alteration in iron homeostasis and regulation in the lung
Although not traditionally viewed as an iron-handling organ, cells in the lung require iron. The lung is continuously exposed to endogenous and exogenous iron (89). Cellular iron mainly comes from endogenous sources like transferrin-bound iron, macrophage recycling, and heme-iron from alveolar hemorrhage (Figure 3). Exogenous iron sources include cigarette smoke (CS), which at 0.06% iron, contributes minimally, and PM2.5, which contains 0.5%–5% to 20%–43% iron, with high levels near industrial sites (90–92). Increases in lung iron from exogenous sources are primarily due to particle-bound iron complexes that promote inflammation, oxidative stress, and macrophage iron retention.
Although systemic HAMP regulates overall iron balance, local lung mechanisms in epithelial cells and AMs fine-tune iron handling and influence disease progression. Dysregulation can disrupt redox balance and hinder lung repair. Iron buildup in the alveolar space, particularly as heme-derived compounds, can compromise barrier integrity and elevate epithelial permeability, underscoring the importance of strict iron regulation in the lung (93, 94). Heme-bound iron induces oxidative stress and creates a prooxidant pool in epithelial lining fluid (ELF) that is exacerbated by CS, hemolysis, or lung damage. In contrast, transferrin and ceruloplasmin in ELF act as antioxidants by binding nonheme iron but do not neutralize heme species–like free heme and hemoglobin (95).
Iron metabolism in the alveolar space plays an additional role in host defense, as AMs and epithelial cells regulate iron supply in response to pathogens. Recently, in vitro studies show that AT2-like cells adjust their iron metabolism based on intracellular versus extracellular pathogens, suggesting a role for AT2 cells in regulating lung-iron during infection (96). AMs modify iron handling through iron sequestration as part of nutritional immunity (97, 98). Dysregulated iron in AMs is associated with impaired bacterial clearance and increased exacerbations of chronic lung disease (97, 98).
AMs are well documented for their role in iron regulation in pathological contexts (99, 100). AMs are involved in iron trafficking, use iron for metabolism in phagocytosis, serve as iron reservoirs, and can be independent of the HAMP/FPN axis (101, 102). In response to LPS, AMs upregulate endogenous HAMP expression, highlighting their ability to influence iron metabolism during inflammation (102). Disruption of the local HAMP/FPN axis causes iron deficiency in AMs, impairing phagocytosis. Thus, intracellular iron serves as a metabolic fuel for efficient phagocytosis in AMs. Notably, in IPF, TfR1-deficient AMs were more abundant and associated with decreased survival (103). Dysregulation of iron metabolism in AMs creates a local iron-rich and oxidative microenvironment. This environment directly affects lung epithelial cells by increasing extracellular iron in the alveolar space that is available for import, altering epithelial iron export activity through HAMP, and triggering inflammatory signaling (99). Paracrine signaling, such as SPP1, promotes cross-talk between AT2 cells and macrophages, driving the recruitment of immune cells and maladaptive cell differentiation (50).
There is less documentation of iron regulation in lung epithelial cells in pathological contexts. Systemic iron dysregulation partly contributes to cellular iron regulation in the lung (51, 104). Notably, point mutations in FPN result in iron accumulation in the alveolar region and thickening of the air-blood barrier with AT2 cell hypertrophy and hyperplasia (51). Global HAMP deficiency alters baseline iron in the lung but does not affect smoke-induced injury (105). HAMP/FPN dysfunction affects the alveolar airspace, but lung epithelial cells can facilitate iron uptake pathways, thereby reducing redox-mediated damage (95, 106). Cellular iron uptake receptors, including TfR1, ZIP8/14, and DMT1, are expressed in the lung epithelium (99, 107, 108). While these importers are found in lung epithelial cells, there is no conclusive evidence to confirm their specific role in maintaining steady-state iron regulation. The mechanisms underlying alternate iron uptake pathways in lung epithelial cells, such as TfR2 and LCN2, remain uncertain. In Tfr2-mutant mice, systemic iron overload leads to significant iron accumulation in the lungs, particularly in the airway basement membrane and tissue macrophages (109), suggesting a role in lung epithelial cells. In COPD, the presence of LCN2 and ferritin in bronchoalveolar fluid (BALF) suggests potential mechanisms for iron absorption (110), with AT2 cells reported to internalize native ferritin (111). Ferritin light chain (FTL) is one of the most abundant transcripts in lung tissue, with both ferritin subunits localizing to alveolar cells (112, 113). However, it remains unclear whether AT2 cells take up iron-bound ferritin and if other lung cell types use similar endocytic mechanisms, as excess iron in lung cells is either stored in ferritin or exported by FPN (114).
Despite limited research, given their unique position at the air-blood barrier, it is likely that AT2 and AT1 cells use multiple iron uptake and transport pathways to regulate iron levels in the alveolar space. Direct regulation of iron in AT1 cells remains underexplored compared with that in AT2 cells. However, AT1 cells are known to be prone to ferroptosis due to oxidative stress. Excess heme-derived iron was shown to be a potent mitochondrial toxin in AT1 cells, impairing mitochondrial metabolism and membrane integrity (94). Moreover, recent work highlights a possible affinity for iron regulatory genes in alveolar repair and alveolar cell identity (115, 116) as well as the association between iron buildup in the alveolar space and restrictive lung disease (104). The alveolar epithelium is vulnerable to such disruptions in iron levels in the alveolar space. Elevated iron levels are associated with alterations in surfactant biophysical properties (117, 118), mitochondrial iron dysregulation in AT2 cells promotes oxidative stress, and age-related changes in iron regulatory gene expression may impair their regenerative capacity (49, 116). Together, these studies further emphasize the important role of iron handling in maintaining alveolar function.
-
Iron as a determinant of alveolar epithelial fate and repair
Numerous lung cells contribute to the development of lung disease; still, there is increasing evidence that iron homeostasis influences AT2 renewal, differentiation, and repair capacity through mitochondrial dysfunction, ferroptosis, and senescence (Figure 4). Given that AT2s are progenitor cells, evidence from broader themes of stem cell research can be informative. A study from Kao and colleagues shows that labile iron levels serve as a rheostat for cellular quiescence in mitotic hematopoietic stem cells (HSCs), suggesting that iron is crucial for determining stem cell fate (119). Variations in cytoplasmic iron levels influence the equilibrium between self-renewal and differentiation in adult stem cells, determining HSC state. Low intracellular iron favors quiescence and self-renewal, preserving long-term regenerative capacity, while high iron triggers metabolic and oxidative stress pathways, pushing HSCs toward differentiation or exhaustion (119).
 Figure 4
Figure 4Role of iron regulation in alveolar repair. A schematic illustrating how iron levels influence cell fate and lung repair in the alveoli. In iron deficiency, alveolar epithelial cells experience metabolic stress, resulting in mitochondrial dysfunction and decreased surfactant production. This stress reduces their capacity to self-renew and differentiate. In alveolar macrophages, stress triggers premature senescence and impairs iron recycling, which weakens phagocytosis and immune responses, leading to immune exhaustion during chronic iron deficiency. Conversely, under normal iron levels, alveolar cells manage iron flux, reducing senescence and ferroptosis, while maintaining immune function. Iron overload causes oxidative stress, mitochondrial dysfunction, ferroptosis, epithelial loss, and tissue damage, impairing regeneration and differentiation. Excess iron in macrophages hampers antimicrobial activity, promotes inflammation and fibrosis, and raises the risk of chronic lung disease. Created in BioRender.
A 2025 study by Zhuang et al. shows that iron homeostasis regulates AT2 cell renewal, differentiation, and lung repair in lung adenocarcinoma cancer models. Aging-related functional iron deficiency in AT2 cells reduces self-renewal and regenerative abilities (52). Although the precise intracellular iron pools and trafficking routes in isolated AT2 cells have not been fully assessed, the study shows that aging rewires iron handling in AT2 cells by inducing nuclear protein 1 (NUPR1), which upregulates LCN2, lowering bioavailable iron, limiting stemness, and inhibiting tumorigenesis. Knocking out the NUPR1/LCN2 axis or iron supplementation in aged AT2 cells can enhance renewal and rescue stemness but also promote tumorigenesis in aged lungs (52). Collectively, these studies emphasize the importance of maintaining an appropriate iron balance for effective AT2 cell–mediated lung repair. Disruptions in iron homeostasis directly impair epithelial cell regeneration, ultimately hindering alveolar repair in chronic lung diseases.
-
Iron dysregulation in chronic lung disease
Iron dysregulation is linked to chronic lung disorders, including IPF and COPD, in which excess iron leads to oxidative stress, ongoing epithelial damage, impaired regeneration, and accelerated disease progression. Exposure to exogenous iron increases susceptibility to respiratory diseases, as seen in individuals working in iron ore mines, who often develop persistent respiratory symptoms even after exposure ends (120).
While iron dysregulation affects lung epithelial repair and lung disease progression in several lung diseases (Table 1), this Review focuses specifically on COPD and IPF. Even though COPD and IPF have distinct pathologies, the role of iron homeostasis in epithelial lung repair is well documented, with evidence of sustained imbalances in iron metabolism. These imbalances compromise AT2 regenerative capacity, promote oxidative stress, induce epithelial ferroptosis (121, 122), and disrupt epithelial barrier integrity, ultimately impairing effective alveolar regeneration and perpetuating disease.
IPF. IPF is a chronic, progressive interstitial lung disease of unknown etiology, characterized by irreversible fibrosis and a rapid decline in lung function (123, 124). Iron accumulation has been observed in lung tissue and BALF of individuals with IPF, with increased iron levels negatively correlating with disease severity (109). Additionally, mitochondrial ROS levels in whole lung tissue from individuals with IPF are substantially increased (125). Iron overload is increasingly linked to the development of fibrosis in the lung and other organs, such as the kidney (109, 126). Genetically, a higher frequency of polymorphisms in the HFE gene, which underlies hereditary hemochromatosis, has been observed in individuals with IPF and is associated with increased iron-dependent ROS generation in BALF-derived cells (127).
BLM administration is a standard experimental model of pulmonary fibrosis; however, the role of iron in BLM-induced injury is complex and context dependent. BLM itself functions as an iron chelator and requires iron to generate ROS and induce DNA damage (128). This property of BLM does not preclude the possibility that exogenous or dysregulated iron levels can modulate the severity of lung injury. In support of this notion, Maus et al. demonstrated that intratracheal administration of iron alone can induce acute lung injury and fibrosis in mice (126). Unfortunately, the literature on the effects of iron in BLM-induced injury is not entirely consistent. For example, systemic iron overload, via a high-iron diet, in HAMP-knockout mice did not exacerbate BLM-induced fibrosis (129), but fibrotic responses were reduced in iron-deficient hamsters subjected to BLM (130). Tfr2-mutant mice, which exhibit systemic iron overload, have markedly worsened fibrosis following intratracheal BLM exposure (109). Treatment with iron chelators at the time of BLM injury onset mitigated lung fibrosis, further implicating iron as a pathogenic factor in this model (53). These findings suggest that the source, compartmentalization, and timing of iron availability — rather than total body iron status — may critically influence disease outcomes. This variability underscores the importance of the experimental context when interpreting iron’s contribution to fibrosis.
As noted above, AMs are the predominant iron+ cells in IPF (131, 132). In the BLM-induced pulmonary fibrosis mouse model, intracellular iron levels are increased not only in AMs, but in AT2 cells and fibroblasts (49, 53, 133, 134). Studies of the alveolar epithelium suggest that cellular and mitochondrial iron accumulation in AT2 cells promote ferroptosis (53) and mitochondrial dysfunction (49), respectively. However, AT2 cell iron status was evaluated in immortal murine AT2-like MLE-12 cells or whole lung tissue, limiting specific conclusions on AT2 cell iron status. Nonetheless, given the high lipid content in AT2 cells and their role in surfactant production, it is plausible that disruptions in AT2 cell iron could heighten lipid peroxidation. Notably, Pei et al. demonstrated that iron chelation and inhibition of ferroptosis with liproxstatin inhibited the development of BLM-induced fibrosis in a mouse model (135). However, the use of intraperitoneal liproxstatin administration does not allow definitive conclusions about the local cell types targeted by the treatment.
Despite emerging research on the role of iron in fibrotic lung remodeling, current knowledge centers on fibroblasts and immune cells (134), with a relatively limited understanding of how iron dysregulation directly affects alveolar epithelial cells, particularly AT2 cells. This represents a critical gap, as AT2 cell dysfunction and failed repair are both hallmarks of IPF. Additional studies are needed to understand how iron-induced cell fates, such as ferroptosis and impaired iron trafficking, compromise alveolar epithelial repair and reveal potential therapeutic targets.
COPD. COPD is characterized by chronic inflammation, airway epithelial remodeling, destruction of alveolar epithelium, and dysfunctional repair processes. The most prominent risk factors for COPD include CS or other inhaled environmental exposures, age, and genetic factors. GWAS have identified significant SNPs in IREB2 (which encodes IRP2), in severe COPD cases, suggesting IREB2 as a COPD susceptibility gene (136). IRP2 levels are increased in lung tissues from patients with COPD (136). In lung epithelial cells, upregulation of IREB2 via circular RNA circSAV1 increased cellular iron and subsequent ferroptosis (137). IRP2-deficient mice are protected against CS exposure–induced experimental COPD as a result of limited mitochondrial iron loading (54) and from developing pulmonary inflammation in a tracheal banding surgery airway obstruction model (138). Porphyria, a genetic disease of dysregulated iron metabolism, is associated with an increased risk of COPD (139).
Systemic iron dysregulation is present in COPD, with elevated intracellular iron deposits detected in lung tissues from patients with very severe COPD (GOLD 4 stage) compared with healthy individuals acting as controls (140). Both current and former smokers exhibit elevated iron and ferritin levels in BALF (90) and AMs (141–144) compared with nonsmoking individuals acting as controls. COPD exhibited an increase in the quantity of iron and percentage of iron+ AMs, and this increase correlated with disease severity and airflow obstruction (144). Within the COPD cohort of the Subpopulations and Intermediate Outcome Measures in COPD Study (SPIROMICS), iron and ferritin were elevated in the BALF of both patients with COPD and smokers without COPD (110). This increase in iron markers in BALF correlated with disease exacerbation risk; however, the same correlation was not observed with plasma markers. Serum levels of HAMP were increased in individuals with mild-to-moderate COPD and those with COPD exacerbations (145, 146); however, HAMP was decreased in severe (GOLD 3/4) COPD (147).
While iron overload appears to be present at the cellular and tissue level in COPD, systemic iron overload is more variable. Several studies have demonstrated higher serum iron, ferritin and LCN2 in individuals with COPD compared with individuals acting as controls (91,146, 148), while other reports have not observed similar changes (149) or demonstrated systemic anemia (89). Studies suggest that systemic iron deficiency is present in a proportion of patients with COPD (10%–40%) (145, 148, 150, 151), despite possible iron overload at the lung tissue or cellular level.
In CS-exposed mice and other experimental COPD models, iron is increased in lung tissue and BALF (81, 138). In mice, exposure to environmental iron powder intensifies lung inflammation, oxidative stress, and remodeling, which are more pronounced when underlying lung damage is present (152). Given the presence of cellular iron overload, ferroptosis has been implicated in COPD pathogenesis, as evidenced in human COPD lung tissues (153) and AT2 cells (154). Additionally, CS exposure in both in vivo and in vitro models induces labile iron accumulation and ferroptotic cell death (122, 155). Furthermore, CS-induced ferroptosis was inhibited both by iron chelators, such as deferoxamine (DFO), and ferroptosis inhibitors, ferrostatin-1 and liproxstatin-1 (122).
Comparisons between COPD and IPF highlight both shared and distinct mechanisms of alveolar injury and repair. In both diseases, iron loading occurs in lung tissue and BALF, with strong evidence of localization to AMs. While ferroptosis signatures have been suggested in AT2 cells, direct evidence in primary cells in either condition is lacking. Both diseases feature transitional epithelial states, with AT2 cells consistently described as senescent, suggesting that iron accumulation and senescence may interact to impair regenerative and repair capacity. While iron regulation shows therapeutic promise, its direct effect on AT2 function remains poorly defined. Clarifying how iron overload alters AT2 behavior over time may uncover intervention points to prevent irreversible lung damage.
-
Therapeutic opportunities targeting iron pathways for lung regeneration
As highlighted in this Review, iron deposition appears to be a key pathological feature of IPF and COPD, with environmental exposures, such as CS, linked to elevated lung iron levels. Genetic factors may further influence iron handling and shift disease susceptibility. Thus, current data support the exploration of therapies that target iron homeostasis.
Iron chelators are compounds (natural or synthetic) that bind iron to reduce iron availability and limit related complications. In the United States, 3 iron chelators are FDA approved: DFO, deferiprone (DFX), and deferasirox (156). These drugs have historically been used to treat iron overload in hepatic, hematologic, and cardiac disease in the absence of anemia (157, 158). DFO is an intravenous agent that chelates extracellular iron and binds to ferritin. While not commonly used to treat COPD, DFO is the standard treatment for acute iron overload (159). DFO has been shown to improve CS-induced inflammation, lung injury, and ferroptosis in lung epithelial cells (54) and reduce downstream ferroptosis in nasal and lung epithelium in CS-induced injury models (122, 160).
In contrast, DFX is orally available and chelates intracellular iron. In a COPD mouse model induced by CS, DFX reduced lung inflammation and injury (54) and reduced iron load and CS-induced ferroptosis in lung epithelial cells (122). These benefits could be achieved through inhaled delivery, providing direct exposure to the lungs. While iron chelation and ferroptosis inhibition have demonstrated benefits in animal models of chronic lung disease, it remains unclear how different cell types contribute to this protection, how lung repair is affected, and how efficient these drugs may be in human disease. Furthermore, there could be different responses in early versus late disease and active versus former smokers with established disease.
The therapeutic efficacy of iron chelators is partly hindered by their short half-life and cytotoxicity. In the cancer field, the use of nanomaterials to prolong half-life, enhance cellular delivery, and increase chelation capacity has been proposed (161, 162). For some iron-related pathways, such as those involving high versus low HAMP in COPD, further studies are needed before targeting them with therapeutic interventions. Additional therapies could target specific components of iron metabolism beyond systemic chelation, such as mitochondrial pathways involved in Fe-S cluster assembly or heme synthesis. Mitochondria-targeted iron chelators are being developed (163), raising important considerations, such as differential effects of modulating cytosolic versus mitochondrial iron pools.
While iron chelation has been proposed as a therapeutic strategy to counter iron overload in the lung, its clinical utility remains complex and highly context dependent. Treatments with iron chelators present particular challenges, as differences in dose, route, and duration of administration can lead to inconsistent or even contradictory outcomes of increased infection risk, exacerbated anemia (89, 164), and impairment of essential cellular processes that rely on iron, including mitochondrial respiration, DNA synthesis, and cellular proliferation. There are studies showing that iron chelators induce mitophagy (165, 166). Iron is central to AT2 cell function and by extension surfactant production, given that surfactant synthesis and secretion rely on AT2-specific lamellar body dynamics and are powered by mitochondrial energy metabolism. Disruption of lipid metabolism in AT2 cells via CS (167) reduces key surfactant phospholipids and increases markers of ferroptosis, highlighting a potential link between iron imbalance, oxidative damage, and impaired AT2 function.
Therefore, approaches that more selectively modulate downstream consequences of iron dysregulation are gaining attention. Interventions targeting cell death pathways downstream of iron, such as ferroptosis, hold promise for these chronic lung diseases (88, 122). Targeting ferroptosis not only addresses iron dysregulation, but may preserve AT2 cell viability and progenitor capacity. Pharmacological blockade of ferroptotic pathways — through lipid peroxidation inhibitors, iron regulators, or GPX4 stabilizers — has the potential to protect AT2 cells from oxidative injury, preserve their self-renewal and differentiation capacity, and support epithelial regeneration. Moving beyond systemic approaches, more targeted strategies — such as localized delivery or combination therapies — may improve efficacy and reduce off-target effects. In this context, emerging approaches such as iron-activated senolytic prodrugs, which selectively exploit elevated iron levels in senescent cells, offer promising avenues for therapy (168). Combining inhaled iron chelators with senolytic agents could represent a future, more precise therapeutic strategy to modulate iron homeostasis and improve outcomes in lung disease.
-
Future directions and gaps
Although iron regulation is critical in chronic lung diseases such as IPF and COPD, many questions still remain. What causes pulmonary iron overload — whether it results from impaired export, excessive uptake, HAMP/FPN dysregulation, or vascular leakage — and how do these mechanisms differ by disease type or stage? Is iron dysregulation a cause or a consequence of the disease? It is unknown whether iron overload accelerates disease or worsens existing conditions, as excessive iron is also naturally linked to aging (169, 170). Additional studies are needed to investigate the roles of iron in both acute and chronic conditions. Diseases like IPF and COPD likely involve ongoing iron accumulation in AT2 cells, whereas acute injuries involve rapid increases in iron and heme due to barrier breakdown, which trigger protective responses, including upregulation of ferritin and HMOX-1. A major limitation in current research is the direct isolation of AT2 cells to investigate their iron metabolism. Although Maus et al. suggested that iron accumulates within AT2 cells, they did not identify the classical pathways typically associated with iron overload (126). As a result, it is still unclear how iron affects the function of healthy AT2 cells, transitional states, or AT1 cells. Existing signatures may be confounded by the heterogeneity of epithelial cell populations rather than reflecting true AT2-specific biology.
Knowledge gaps extend beyond the alveolar epithelium; other lung regions with progenitors, such as basal, club, and bronchioalveolar stem cells, may be affected by iron dysregulation. Advanced techniques like single-cell transcriptomics can help identify vulnerable cell populations. Understanding these time- and cell-specific changes is key to developing targeted therapies, as strategies effective in acute settings could be harmful in chronic diseases. Filling these gaps is essential for creating precise treatments that enhance iron regulation, support alveolar repair, and slow lung disease progression.
-
Conflict of interest
SMC is supported by a Research Ireland Future Research Leaders (FRL4862) award and a Research Ireland Laureate Award (IRCLA/2022/3619). SMC holds a patent (US 10,905,682 B2) for the use of mitochondrial iron chelators for the treatment of COPD.
-
Funding support
This work is the result of NIH funding, in whole or in part, and is subject to the NIH Public Access Policy. Through acceptance of this federal funding, the NIH has been given a right to make the work publicly available in PubMed Central.
- NIH National Heart, Lung, and Blood Institute (R01HL168050) to CRK.
- Burroughs Wellcome Fund Career Award for Medical Scientists to CRK.
- Research Ireland Future Research Leaders (FRL4862) award to SMC.
- Research Ireland Laureate Award (IRCLA/2022/3619) to SMC.
-
Footnotes
Copyright: © 2026, Mbaekwe et al. This is an open access article published under the terms of the Creative Commons Attribution 4.0 International License.
Reference information: JCI Insight. 2026;11(9):e199953. https://doi.org/10.1172/jci.insight.199953.
-
References
- Hsia CCW, et al. Lung structure and the intrinsic challenges of gas exchange. Compr Physiol. 2016;6(2):827–895.
- Wang Y, et al. Repair and regeneration of the alveolar epithelium in lung injury. FASEB J. 2024;38(8):e23612.
- Kim KJ, Malik AB. Protein transport across the lung epithelial barrier. Am J Physiol Lung Cell Mol Physiol. 2003;284(2):L247–L259.
- Song L, et al. Cell cross-talk in alveolar microenvironment: from lung injury to fibrosis. Am J Respir Cell Mol Biol. 2024;71(1):30–42.
- Gillich A, et al. Capillary cell-type specialization in the alveolus. Nature. 2020;586(7831):785–789.
- Ushakumary MG, et al. Resident interstitial lung fibroblasts and their role in alveolar stem cell niche development, homeostasis, injury, and regeneration. Stem Cells Transl Med. 2021;10(7):1021–1032.
- Westphalen K, et al. Sessile alveolar macrophages communicate with alveolar epithelium to modulate immunity. Nature. 2014;506(7489):503–506.
- Han W, et al. Identification and characterization of alveolar and recruited lung macrophages during acute lung inflammation. J Immunol. 2023;210(11):1827–1836.
- Barkauskas CE, et al. Type 2 alveolar cells are stem cells in adult lung. J Clin Invest. 2013;123(7):3025–3036.
- Evans MJ, et al. Transformation of alveolar type 2 cells to type 1 cells following exposure to NO2. Exp Mol Pathol. 1975;22(1):142–150.
- Basil MC, et al. Lung repair and regeneration: advanced models and insights into human disease. Cell Stem Cell. 2024;31(4):439–454.
- Brownfield DG, et al. Alveolar cell fate selection and lifelong maintenance of AT2 cells by FGF signaling. Nat Commun. 2022;13(1):7137.
- Nabhan AN, et al. Single-cell Wnt signaling niches maintain stemness of alveolar type 2 cells. Science. 2018;359(6380):1118–1123.
- Basil MC, et al. The cellular and physiological basis for lung repair and regeneration: past, present, and future. Cell Stem Cell. 2020;26(4):482–502.
- Zacharias WJ, et al. Regeneration of the lung alveolus by an evolutionarily conserved epithelial progenitor. Nature. 2018;555(7695):251–255.
- Toth A, et al. Alveolar epithelial progenitor cells require Nkx2-1 to maintain progenitor-specific epigenomic state during lung homeostasis and regeneration. Nat Commun. 2023;14(1):8452.
- Hassan D, Chen J. CEBPA restricts alveolar type 2 cell plasticity during development and injury-repair. Nat Commun. 2024;15(1):4148.
- Finn J, et al. Dlk1-mediated temporal regulation of Notch signaling is required for differentiation of alveolar type II to type I cells during repair. Cell Rep. 2019;26(11):2942–2954.
- Liu Z, et al. MAPK-mediated YAP activation controls mechanical-tension-induced pulmonary alveolar regeneration. Cell Rep. 2016;16(7):1810–1819.
- LaCanna R, et al. Yap/Taz regulate alveolar regeneration and resolution of lung inflammation. J Clin Invest. 2019;129(5):2107–2122.
- DiGiovanni GT, et al. Epithelial Yap/Taz are required for functional alveolar regeneration following acute lung injury. JCI Insight. 2023;8(19):e173374.
- Zhang J, Liu Y. Epithelial stem cells and niches in lung alveolar regeneration and diseases. Chin Med J Pulm Crit Care Med. 2024;2(1):17–26.
- Juul NH, et al. Niche cells and signals that regulate lung alveolar stem cells in vivo. Cold Spring Harb Perspect Biol. 2020;12(12):a035717.
- Yao C, et al. Senescence of alveolar type 2 cells drives progressive pulmonary fibrosis. Am J Respir Crit Care Med. 2021;203(6):707–717.
- Nureki SI, et al. Expression of mutant Sftpc in murine alveolar epithelia drives spontaneous lung fibrosis. J Clin Invest. 2018;128(9):4008–4024.
- Enomoto Y, et al. Autocrine TGF-β-positive feedback in profibrotic AT2-lineage cells plays a crucial role in non-inflammatory lung fibrogenesis. Nat Commun. 2023;14(1):4956.
- Panos RJ, et al. Keratinocyte growth factor and hepatocyte growth factor/scatter factor are heparin-binding growth factors for alveolar type II cells in fibroblast-conditioned medium. J Clin Invest. 1993;92(2):969–977.
- Yuan T, et al. FGF10-FGFR2B signaling generates basal cells and drives alveolar epithelial regeneration by bronchial epithelial stem cells after lung injury. Stem Cell Reports. 2019;12(5):1041–1055.
- Zhao L, et al. Transdifferentiation of alveolar epithelial type II to type I cells is controlled by opposing TGF-β and BMP signaling. Am J Physiol Lung Cell Mol Physiol. 2013;305(6):L409–L418.
- Cakarova L, et al. Macrophage tumor necrosis factor-alpha induces epithelial expression of granulocyte-macrophage colony-stimulating factor: impact on alveolar epithelial repair. Am J Respir Crit Care Med. 2009;180(6):521–532.
- Choi J, et al. Inflammatory signals induce AT2 cell-derived damage-associated transient progenitors that mediate alveolar regeneration. Cell Stem Cell. 2020;27(3):366–382.
- Cloonan SM, Choi AMK. Mitochondria in lung disease. J Clin Invest. 2016;126(3):809–820.
- Massaro GD, et al. Lung oxygen consumption and mitochondria of alveolar epithelial and endothelial cells. J Appl Physiol. 1975;38(4):588–592.
- Ali M, et al. MICU1-dependent mitochondrial calcium uptake regulates lung alveolar type 2 cell plasticity and lung regeneration. JCI Insight. 2022;7(4):e154447.
- Wang S, et al. Glutamine metabolism is required for alveolar regeneration during lung injury. Biomolecules. 2022;12(5):728.
- Wang Z, et al. Enhanced glycolysis-mediated energy production in alveolar stem cells is required for alveolar regeneration. Cell Stem Cell. 2023;30(8):1028–1042.
- Angeles-Lopez QD, et al. Regulation of lung progenitor plasticity and repair by fatty acid oxidation. JCI Insight. 2025;10(3):e165837.
- Han S, et al. Mitochondrial integrated stress response controls lung epithelial cell fate. Nature. 2023;620(7975):890–897.
- Liu K, et al. Tracing the origin of alveolar stem cells in lung repair and regeneration. Cell. 2024;187(10):2428–2445.
- Bi W, et al. Alveolar epithelial type 2 cells in acute lung injury and acute respiratory distress syndrome: spatiotemporal fate and regulation. Burns Trauma. 2025;13:tkaf050.
- Wang F, et al. Regulation of epithelial transitional states in murine and human pulmonary fibrosis. J Clin Invest. 2023;133(22):e165612.
- Strunz M, et al. Alveolar regeneration through a Krt8+ transitional stem cell state that persists in human lung fibrosis. Nat Commun. 2020;11(1):3559.
- Hernandez-Segura A, et al. Hallmarks of cellular senescence. Trends Cell Biol. 2018;28(6):436–453.
- Kumar PA, et al. Distal airway stem cells yield alveoli in vitro and during lung regeneration following H1N1 influenza infection. Cell. 2011;147(3):525–538.
- Lv Z, et al. Alveolar regeneration by airway secretory-cell-derived p63+ progenitors. Cell Stem Cell. 2024;31(11):1685–1700.
- Fernanda de Mello Costa M, et al. Basal-like progenitor cells: a review of dysplastic alveolar regeneration and remodeling in lung repair. Stem Cell Reports. 2020;15(5):1015–1025.
- Su W, et al. YAP1 inhibits the senescence of alveolar epithelial cells by targeting Prdx3 to alleviate pulmonary fibrosis. Exp Mol Med. 2024;56(7):1643–1654.
- Abbaspour N, et al. Review on iron and its importance for human health. J Res Med Sci. 2014;19(2):164–174.View this article via: PubMed Google Scholar
- Shao M, et al. Abnormal mitochondrial iron metabolism damages alveolar type II epithelial cells involved in bleomycin-induced pulmonary fibrosis. Theranostics. 2024;14(7):2687–2705.
- Du X, et al. SPP1 regulates alveolar type 2 cell-macrophage cross talk and epithelial cell fate in iron-driven lung fibrosis. Am J Physiol Cell Physiol. 2025;329(3):C779–C800.
- Mühlfeld C, et al. Air-blood barrier thickening and alterations of alveolar epithelial type 2 cells in mouse lungs with disrupted hepcidin/ferroportin regulatory system. Histochem Cell Biol. 2019;151(3):217–228.
- Zhuang X, et al. Ageing limits stemness and tumorigenesis by reprogramming iron homeostasis. Nature. 2025;637(8044):184–194.
- Cheng H, et al. Iron deposition-induced ferroptosis in alveolar type II cells promotes the development of pulmonary fibrosis. Biochim Biophys Acta Mol Basis Dis. 2021;1867(12):166204.
- Cloonan SM, et al. Mitochondrial iron chelation ameliorates cigarette smoke-induced bronchitis and emphysema in mice. Nat Med. 2016;22(2):163–174.
- Andreini C, et al. The human iron-proteome. Metallomics. 2018;10(9):1223–1231.
- Chevion M. A site-specific mechanism for free radical induced biological damage: the essential role of redox-active transition metals. Free Radic Biol Med. 1988;5(1):27–37.
- Lane DJR, et al. Duodenal cytochrome b (DCYTB) in iron metabolism: an update on function and regulation. Nutrients. 2015;7(4):2274–2296.
- Shayeghi M, et al. Identification of an intestinal heme transporter. Cell. 2005;122(5):789–801.
- Raffin SB, et al. Intestinal absorption of hemoglobin iron-heme cleavage by mucosal heme oxygenase. J Clin Invest. 1974;54(6):1344–1352.
- Poss KD, Tonegawa S. Heme oxygenase 1 is required for mammalian iron reutilization. Proc Natl Acad Sci U S A. 1997;94(20):10919–10924.
- Jandl JH, Katz JH. The plasma-to-cell cycle of transferrin. J Clin Invest. 1963;42(3):314–326.
- Bratosin D, et al. Cellular and molecular mechanisms of senescent erythrocyte phagocytosis by macrophages. A review. Biochimie. 1998;80(2):173–195.
- Drakesmith H, Prentice AM. Hepcidin and the iron-infection axis. Science. 2012;338(6108):768–772.
- Nemeth E, et al. Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science. 2004;306(5704):2090–2093.
- Harding C, et al. Receptor-mediated endocytosis of transferrin and recycling of the transferrin receptor in rat reticulocytes. J Cell Biol. 1983;97(2):329–339.
- Ohgami RS, et al. Identification of a ferrireductase required for efficient transferrin-dependent iron uptake in erythroid cells. Nat Genet. 2005;37(11):1264–1269.
- Wang CY, et al. ZIP8 is an iron and zinc transporter whose cell-surface expression is up-regulated by cellular iron loading. J Biol Chem. 2012;287(41):34032–34043.
- Jenkitkasemwong S, et al. Physiologic implications of metal-ion transport by ZIP14 and ZIP8. Biometals. 2012;25(4):643–655.
- Bachman MA, et al. Interaction of lipocalin 2, transferrin, and siderophores determines the replicative niche of Klebsiella pneumoniae during pneumonia. mBio. 2012;3(6):e00224-11.
- Shi H, et al. A cytosolic iron chaperone that delivers iron to ferritin. Science. 2008;320(5880):1207–1210.
- Chen W, et al. Abcb10 physically interacts with mitoferrin-1 (Slc25a37) to enhance its stability and function in the erythroid mitochondria. Proc Natl Acad Sci U S A. 2009;106(38):16263–16268.
- Paradkar PN, et al. Regulation of mitochondrial iron import through differential turnover of mitoferrin 1 and mitoferrin 2. Mol Cell Biol. 2009;29(4):1007–1016.
- Quigley JG, et al. Identification of a human heme exporter that is essential for erythropoiesis. Cell. 2004;118(6):757–766.
- Keel SB, et al. A heme export protein is required for red blood cell differentiation and iron homeostasis. Science. 2008;319(5864):825–828.
- Mastrogiannaki M, et al. HIF-2alpha, but not HIF-1alpha, promotes iron absorption in mice. J Clin Invest. 2009;119(5):1159–1166.
- Moroishi T, et al. HERC2 targets the iron regulator FBXL5 for degradation and modulates iron metabolism. J Biol Chem. 2014;289(23):16430–16441.
- Hognon C, et al. The iron maiden. Cytosolic aconitase/IRP1 conformational transition in the regulation of ferritin translation and iron hemostasis. Biomolecules. 2021;11(9):1329.
- Anderson SA, et al. The IRP1-HIF-2α axis coordinates iron and oxygen sensing with erythropoiesis and iron absorption. Cell Metab. 2013;17(2):282–290.
- Terzi EM, et al. Iron-sulfur cluster deficiency can be sensed by IRP2 and regulates iron homeostasis and sensitivity to ferroptosis independent of IRP1 and FBXL5. Sci Adv. 2021;7(22):eabg4302.
- Galy B, et al. Mechanisms controlling cellular and systemic iron homeostasis. Nat Rev Mol Cell Biol. 2024;25(2):133–155.
- Mancias JD, et al. Quantitative proteomics identifies NCOA4 as the cargo receptor mediating ferritinophagy. Nature. 2014;509(7498):105–109.
- Anandhan A, et al. NRF2 controls iron homeostasis and ferroptosis through HERC2 and VAMP8. Sci Adv. 2023;9(5):eade9585.
- Katona G, et al. Raman-assisted crystallography reveals end-on peroxide intermediates in a nonheme iron enzyme. Science. 2007;316(5823):449–453.
- Chen Y, et al. Oxidative stress induces mitochondrial iron overload and ferroptotic cell death. Sci Rep. 2023;13(1):15515.
- Paul BT, et al. Mitochondria and iron: current questions. Expert Rev Hematol. 2017;10(1):65–79.
- Saito Y. Lipid peroxidation products as a mediator of toxicity and adaptive response - the regulatory role of selenoprotein and vitamin E. Arch Biochem Biophys. 2021;703:108840.
- Dixon SJ, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060–1072.
- Yang L, et al. Targeting ferroptosis as a vulnerability in pulmonary diseases. Cell Death Dis. 2022;13(7):649.
- Cloonan SM, et al. The “iron”-y of iron overload and iron deficiency in chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2017;196(9):1103–1112.
- Ghio AJ, et al. Particulate matter in cigarette smoke alters iron homeostasis to produce a biological effect. Am J Respir Crit Care Med. 2008;178(11):1130–1138.
- Zhang WZ, et al. Smoking-induced iron dysregulation in the lung. Free Radic Biol Med. 2019;133:238–247.
- O’Day PA, et al. Iron speciation in respirable particulate matter and implications for human health. Environ Sci Technol. 2022;56(11):7006–7016.
- Shaver CM, et al. Cell-free hemoglobin: a novel mediator of acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2016;310(6):L532–L541.
- Chintagari NR, et al. Oxidized ferric and ferryl forms of hemoglobin trigger mitochondrial dysfunction and injury in alveolar type I cells. Am J Respir Cell Mol Biol. 2016;55(2):288–298.
- Pacht ER, Davis WB. Role of transferrin and ceruloplasmin in antioxidant activity of lung epithelial lining fluid. J Appl Physiol (1985). 1988;64(5):2092–2099.
- Grubwieser P, et al. Airway epithelial cells differentially adapt their iron metabolism to infection with Klebsiella pneumoniae and Escherichia coli in vitro. Front Cell Infect Microbiol. 2022;12:875543.
- Johnson EE, Wessling-Resnick M. Iron metabolism and the innate immune response to infection. Microbes Infect. 2012;14(3):207–216.
- Cherayil BJ. The role of iron in the immune response to bacterial infection. Immunol Res. 2011;50(1):1–9.
- Neves J, et al. Iron Homeostasis in the lungs-a balance between health and disease. Pharmaceuticals (Basel). 2019;12(1):5.
- Soares MP, Hamza I. Macrophages and iron metabolism. Immunity. 2016;44(3):492–504.
- Winn NC, Volk KM, Hasty AH. Regulation of tissue iron homeostasis: the macrophage “ferrostat. ” JCI Insight. 2020;5(2):e132964.
- Nguyen NB, et al. Hepcidin expression and iron transport in alveolar macrophages. Am J Physiol Lung Cell Mol Physiol. 2006;291(3):L417–L425.
- Allden SJ, et al. The transferrin receptor CD71 delineates functionally distinct airway macrophage subsets during idiopathic pulmonary fibrosis. Am J Respir Crit Care Med. 2019;200(2):209–219.
- Neves J, et al. Disruption of the hepcidin/ferroportin regulatory system causes pulmonary iron overload and restrictive lung disease. EBioMedicine. 2017;20:230–239.
- Perez E, et al. Hepcidin is essential for alveolar macrophage function and is disrupted by smoke in a murine chronic obstructive pulmonary disease model. J Immunol. 2020;205(9):2489–2498.
- Gutteridge JM, et al. Pro-oxidant iron is present in human pulmonary epithelial lining fluid: implications for oxidative stress in the lung. Biochem Biophys Res Commun. 1996;220(3):1024–1027.
- Brain JD, et al. Effects of iron status on transpulmonary transport and tissue distribution of Mn and Fe. Am J Respir Cell Mol Biol. 2006;34(3):330–337.
- Turi JL, et al. Duodenal cytochrome b: a novel ferrireductase in airway epithelial cells. Am J Physiol Lung Cell Mol Physiol. 2006;291(2):L272–L280.
- Ali MK, et al. Critical role for iron accumulation in the pathogenesis of fibrotic lung disease. J Pathol. 2020;251(1):49–62.
- Zhang WZ, et al. Increased airway iron parameters and risk for exacerbation in COPD: an analysis from SPIROMICS. Sci Rep. 2020;10(1):10562.
- Williams MC. Endocytosis in alveolar type II cells: effect of charge and size of tracers. Proc Natl Acad Sci U S A. 1984;81(19):6054–6058.
- Lindskog C, et al. The lung-specific proteome defined by integration of transcriptomics and antibody-based profiling. FASEB J. 2014;28(12):5184–5196.
- Du Y, et al. “LungGENS”: a web-based tool for mapping single-cell gene expression in the developing lung. Thorax. 2015;70(11):1092–1094.
- Yang F, et al. Apical location of ferroportin 1 in airway epithelia and its role in iron detoxification in the lung. Am J Physiol Lung Cell Mol Physiol. 2005;289(1):L14–L23.
- Jia H, et al. A single-cell atlas of lung homeostasis reveals dynamic changes during development and aging. Commun Biol. 2024;7(1):427.
- Zhuang X, Wang Q, Joost S, et al. Aging limits stemness and tumorigenesis in the lung by reprogramming iron homeostasis [preprint]. https://doi.org/10.1101/2024.06.23.600305 Posted on bioRxiv June 28, 2024.
- Hallman M, et al. Transferrin modifies surfactant responsiveness in acute respiratory failure: role of iron-free transferrin as an antioxidant. Pediatr Pulmonol. 1996;22(1):14–22.
- Hallman M, et al. Interaction of transferrin saturated with iron with lung surfactant in respiratory failure. J Appl Physiol (1985). 1994;77(2):757–766.
- Kao YR, et al. An iron rheostat controls hematopoietic stem cell fate. Cell Stem Cell. 2024;31(3):378–397.
- Hedlund U, et al. Persistence of respiratory symptoms in ex-underground iron ore miners. Occup Med (Lond). 2006;56(6):380–385.
- Hu Y, et al. Emerging roles of ferroptosis in pulmonary fibrosis: current perspectives, opportunities and challenges. Cell Death Discov. 2024;10(1):301.
- Yoshida M, et al. Involvement of cigarette smoke-induced epithelial cell ferroptosis in COPD pathogenesis. Nat Commun. 2019;10(1):3145.
- No authors listed. Idiopathic pulmonary fibrosis: diagnosis and treatment. International consensus statement. American Thoracic Society (ATS), and the European Respiratory Society (ERS). Am J Respir Crit Care Med. 2000;161(2 pt 1):646–664.
- Raghu G, et al. Idiopathic pulmonary fibrosis in US Medicare beneficiaries aged 65 years and older: incidence, prevalence, and survival, 2001-11. Lancet Respir Med. 2014;2(7):566–572.
- Jaeger VK, et al. Mitochondrial DNA mutations and respiratory chain dysfunction in idiopathic and connective tissue disease-related lung fibrosis. Sci Rep. 2019;9(1):5500.
- Maus M, et al. Iron accumulation drives fibrosis, senescence and the senescence-associated secretory phenotype. Nat Metab. 2023;5(12):2111–2130.
- Sangiuolo F, et al. HFE gene variants and iron-induced oxygen radical generation in idiopathic pulmonary fibrosis. Eur Respir J. 2015;45(2):483–490.
- Hay J, et al. Mechanisms of bleomycin-induced lung damage. Arch Toxicol. 1991;65(2):81–94.
- Zhang V, et al. Lung iron overload does not exacerbate the fibrotic response to bleomycin in a mouse model of pulmonary fibrosis. Am J Respir Cell Mol Biol. 2020;63(5):713–716.
- Chandler DB, et al. Effect of iron deficiency on bleomycin-induced lung fibrosis in the hamster. Am Rev Respir Dis. 1988;137(1):85–89.
- Lee J, et al. Bronchoalveolar lavage (BAL) cells in idiopathic pulmonary fibrosis express a complex pro-inflammatory, pro-repair, angiogenic activation pattern, likely associated with macrophage iron accumulation. PLoS One. 2018;13(4):e0194803.
- Puxeddu E, et al. Iron laden macrophages in idiopathic pulmonary fibrosis: the telltale of occult alveolar hemorrhage? Pulm Pharmacol Ther. 2014;28(1):35–40.
- Zhu Y, et al. Clioquinol attenuates pulmonary fibrosis through inactivation of fibroblasts via iron chelation. Am J Respir Cell Mol Biol. 2021;65(2):189–200.
- Li Y, et al. Iron-laden macrophage-mediated paracrine profibrotic signaling induces lung fibroblast activation. Am J Physiol Cell Physiol. 2024;327(4):C979–C993.
- Pei Z, et al. Inhibition of ferroptosis and iron accumulation alleviates pulmonary fibrosis in a bleomycin model. Redox Biol. 2022;57:102509.
- DeMeo DL, et al. Integration of genomic and genetic approaches implicates IREB2 as a COPD susceptibility gene. Am J Hum Genet. 2009;85(4):493–502.
- Xia H, et al. N6-Methyladenosine-modified circSAV1 triggers ferroptosis in COPD through recruiting YTHDF1 to facilitate the translation of IREB2. Cell Death Differ. 2023;30(5):1293–1304.
- Toumpanakis D, Vassilakopoulou V, Gu W, et al. Iron Regulatory Protein 2 (IRP2) Deficiency Is Protective Against Resistive Breathing-induced Pulmonary Inflammation. Am J Respir Cell Mol Biol. 2025;73(1):96–108.
- Linet MS, et al. Primary liver cancer, other malignancies, and mortality risks following porphyria: a cohort study in Denmark and Sweden. Am J Epidemiol. 1999;149(11):1010–1015.
- Najafinobar N, et al. ToF-SIMS mediated analysis of human lung tissue reveals increased iron deposition in COPD (GOLD IV) patients. Sci Rep. 2019;9(1):10060.
- Thompson AB, et al. Lower respiratory tract iron burden is increased in association with cigarette smoking. J Lab Clin Med. 1991;117(6):493–499.View this article via: PubMed Google Scholar
- Wesselius LJ, et al. Increased release of ferritin and iron by iron-loaded alveolar macrophages in cigarette smokers. Am J Respir Crit Care Med. 1994;150(3):690–695.
- Wesselius LJ, et al. Alveolar macrophage content of isoferritins and transferrin. Comparison of nonsmokers and smokers with and without chronic airflow obstruction. Am Rev Respir Dis. 1992;145(2 pt 1):311–316.
- Philippot Q, et al. Increased iron sequestration in alveolar macrophages in chronic obstructive pulmonary disease. PLoS One. 2014;9(5):e96285.
- Nickol AH, et al. A cross-sectional study of the prevalence and associations of iron deficiency in a cohort of patients with chronic obstructive pulmonary disease. BMJ Open. 2015;5(7):e007911.
- Tandara L, et al. Systemic inflammation up-regulates serum hepcidin in exacerbations and stabile chronic obstructive pulmonary disease. Clin Biochem. 2015;48(18):1252–1257.
- Duru S, et al. Hepcidin: a useful marker in chronic obstructive pulmonary disease. Ann Thorac Med. 2012;7(1):31–35.
- Pizzini A, et al. The significance of iron deficiency and anemia in a real-life COPD cohort. Int J Med Sci. 2020;17(14):2232–2239.
- Lee CH, et al. Association of serum ferritin levels with smoking and lung function in the Korean adult population: analysis of the fourth and fifth Korean National Health and Nutrition Examination Survey. Int J Chron Obstruct Pulmon Dis. 2016;11:3001–3006.
- Kunitomo Y, et al. Iron deficiency and all-cause hospitalization risk in a clinical cohort of COPD. Chronic Obstr Pulm Dis. 2025;12(1):72–81.
- Plesner LL, et al. Iron deficiency in COPD associates with increased pulmonary artery pressure estimated by echocardiography. Heart Lung Circ. 2017;26(1):101–104.
- Galli TT, et al. Effects of environmental exposure to iron powder on healthy and elastase-exposed mice. Sci Rep. 2024;14(1):9134.
- Günes Günsel G, et al. The arginine methyltransferase PRMT7 promotes extravasation of monocytes resulting in tissue injury in COPD. Nat Commun. 2022;13(1):1303.
- Ma L, et al. Ferroptotic alveolar epithelial type II cells drive TH2 and TH17 mixed asthma triggered by birch pollen allergen Bet v 1. Cell Death Discov. 2024;10(1):96.
- Zeng Z, et al. DNA dioxygenases TET2 deficiency promotes cigarette smoke induced chronic obstructive pulmonary disease by inducing ferroptosis of lung epithelial cell. Redox Biol. 2023;67:102916.
- Mobarra N, et al. A review on iron chelators in treatment of iron overload syndromes. Int J Hematol Oncol Stem Cell Res. 2016;10(4):239–247.View this article via: PubMed Google Scholar
- Junqueira FP, et al. Right and left ventricular function and myocardial scarring in adult patients with sickle cell disease: a comprehensive magnetic resonance assessment of hepatic and myocardial iron overload. J Cardiovasc Magn Reson. 2013;15(1):83.
- Hassan MAM, Tolba OA. Iron chelation monotherapy in transfusion-dependent beta-thalassemia major patients: a comparative study of deferasirox and deferoxamine. Electron Physician. 2016;8(5):2425–2431.
- Borella E, et al. Characterisation of individual ferritin response in patients receiving chelation therapy. Br J Clin Pharmacol. 2022;88(8):3683–3694.
- Pan L, et al. Cigarette smoke-induced epithelial cell ferroptosis promotes neutrophilic inflammation in patients with nasal polyps. J Allergy Clin Immunol. 2025;55(6):1882–1897.
- Kang H, et al. Renal clearable nanochelators for iron overload therapy. Nat Commun. 2019;10(1):5134.
- Duenas-Ramirez P, et al. Highly chelating stellate mesoporous silica nanoparticles for specific iron removal from biological media. J Colloid Interface Sci. 2020;579:140–151.
- Cilibrizzi A, et al. The synthesis and properties of mitochondrial targeted iron chelators. Biometals. 2023;36(2):321–337.
- Parrow NL, et al. Sequestration and scavenging of iron in infection. Infect Immun. 2013;81(10):3503–3514.
- Hara Y, et al. Iron loss triggers mitophagy through induction of mitochondrial ferritin. EMBO Rep. 2020;21(11):e50202.
- Brogyanyi T, et al. Iron chelators as mitophagy agents: Potential and limitations. Biomed Pharmacother. 2024;179:117407.
- Fan LC, et al. Alveolar type II epithelial cell FASN maintains lipid homeostasis in experimental COPD. JCI Insight. 2023;8(16):e163403.
- Admasu TD, et al. Selective ablation of primary and paracrine senescent cells by targeting iron dyshomeostasis. Cell Rep. 2023;42(2):112058.
- Zeidan RS, et al. Iron homeostasis and organismal aging. Ageing Res Rev. 2021;72:101510.
- Xu J, et al. Impaired iron status in aging research. Int J Mol Sci. 2012;13(2):2368–2386.
- Cui Y, et al. Imbalanced and unchecked: the role of metal dyshomeostasis in driving COPD progression. COPD. 2024;21(1):2322605.
- Shen C, et al. PCR array analysis reveals a novel expression profile of ferroptosis-related genes in idiopathic pulmonary fibrosis. BMC Pulm Med. 2025;25(1):98.
- Jiang S, et al. Iron dysregulation and ferroptosis are associated with pulmonary fibrosis: insight from idiopathic pulmonary fibrosis, systemic sclerosis, and COVID-19 patients. J Trace Elem Med Biol. 2025;91:127728.
- Yao M, et al. Ferroptosis: a key driver and therapeutic target in the pathogenesis of acute respiratory distress syndrome. Front Immunol. 2025;16:1567980.
- Ma W, et al. Advances in acute respiratory distress syndrome: focusing on heterogeneity, pathophysiology, and therapeutic strategies. Signal Transduct Target Ther. 2025;10(1):75.
- Ghio AJ, et al. Iron and iron-related proteins in the lower respiratory tract of patients with acute respiratory distress syndrome. Crit Care Med. 2003;31(2):395–400.
- Michels KR, et al. Hepcidin-mediated iron sequestration protects against bacterial dissemination during pneumonia. JCI Insight. 2017;2(6):e92002.
- Ong CLY, et al. Interplay between manganese and iron in pneumococcal pathogenesis: role of the orphan response regulator RitR. Infect Immun. 2013;81(2):421–429.
- Ghio AJ, et al. Iron accumulates in the lavage and explanted lungs of cystic fibrosis patients. J Cyst Fibros. 2013;12(4):390–398.
- Lobbes H, et al. Improvement of iron status with elexacaftor tezacaftor ivacaftor therapy is associated with the correction of systemic inflammation and improvement of lung function: a one-year prospective study. Sci Rep. 2025;15(1):17394.
- Reid DW, et al. Role of lung iron in determining the bacterial and host struggle in cystic fibrosis. Am J Physiol Lung Cell Mol Physiol. 2009;297(5):L795–L802.
- Lelièvre P, et al. Iron dysregulation in human cancer: altered metabolism, biomarkers for diagnosis, prognosis, monitoring and rationale for therapy. Cancers (Basel). 2020;12(12):3524.
- Hinokuma H, et al. Distinct functions between ferrous and ferric iron in lung cancer cell growth. Cancer Sci. 2023;114(11):4355–4364.
- Kuang Y, et al. Iron-dependent CDK1 activity promotes lung carcinogenesis via activation of the GP130/STAT3 signaling pathway. Cell Death Dis. 2019;10(4):297.
- Hanson AL, et al. Iron dysregulation and inflammatory stress erythropoiesis associates with long-term outcome of COVID-19. Nat Immunol. 2024;25(3):471–482.
- Sonnweber T, et al. Persisting alterations of iron homeostasis in COVID-19 are associated with non-resolving lung pathologies and poor patients’ performance: a prospective observational cohort study. Respir Res. 2020;21(1):276.
- Qiu B, et al. Fatal COVID-19 pulmonary disease involves ferroptosis. Nat Commun. 2024;15(1):3816.
- Brigham EP, et al. Iron status is associated with asthma and lung function in US women. PLoS One. 2015;10(2):e0117545.
- Hurrell BP, et al. Iron controls the development of airway hyperreactivity by regulating ILC2 metabolism and effector function. Sci Transl Med. 2024;16(746):eadk4728.
- Ali MK, et al. Crucial role for lung iron level and regulation in the pathogenesis and severity of asthma. Eur Respir J. 2020;55(4):1901340.
- Ghio AJ, et al. Calcium oxalate and iron accumulation in sarcoidosis. Sarcoidosis Vasc Diffuse Lung Dis. 2000;17(2):140–150.View this article via: PubMed Google Scholar
-
Version history
- Version 1 (May 8, 2026): Electronic publication
Article tools
-
View PDF

- Download citation information
- Send a comment
- Terms of use
- Standard abbreviations
- Need help? Email the journal
Metrics
Go to
- Top
- Abstract
- The alveolar epithelium
- Alveolar epithelium renewal and repair processes
- Dysregulation of alveolar renewal and repair
- Iron homeostasis and regulation
- Alteration in iron homeostasis and regulation in the lung
- Iron as a determinant of alveolar epithelial fate and repair
- Iron dysregulation in chronic lung disease
- Therapeutic opportunities targeting iron pathways for lung regeneration
- Future directions and gaps
- Conflict of interest
- Funding support
- Footnotes
- References
- Version history








